Ionizing Radiation and Pediatric X-ray Assessment in Rivers State, Nigeria
| Received 15 Nov, 2025 |
Accepted 10 Feb, 2026 |
Published 20 Feb, 2026 |
Background and Objective: Pediatric populations in Nigeria are highly radiosensitive, yet comprehensive assessments of background ionizing radiation (BIR) and X-ray exposure are scarce, particularly in sub-Saharan Africa. This study evaluated BIR and pediatric X-ray exposure risks at Rivers State University Teaching Hospital, Nigeria, to identify gaps relative to international safety standards and inform protective measures. Materials and Methods: A dual-phase cross-sectional study was conducted. BIR was measured at 37 hospital sites using calibrated Digilert-200 survey meters with GPS localization. Pediatric dosimetry for 39 patients (aged 3-15 years) undergoing routine X-rays was captured using optically stimulated luminescence dosimeters. Key dosimetric indices entrance surface dose (ESD), absorbed dose, effective dose, and excess lifetime cancer risk (ELCR) were calculated using ICRP coefficients and Monte Carlo derived factors. Data were analyzed using SPSS v25; Pearson’s correlation assessed relationships between kVp, mAs, FSD, and ESD, compared with IAEA pediatric DRLs, with p<0.05 considered significant. Results: The BIR exceeded ICRP thresholds in 55% of indoor sites, peaking at 0.020±0.002 mR/hr near radioactive waste storage. Outdoor levels ranged from 0.008±0.002 to 0.020±0.002 mR/hr (mean 0.011±0.003 mR/hr). Pediatric ESDs varied by procedure: Chest X-rays averaged 0.009 mGy, skull 0.368 mGy, and abdomen 0.371 mGy. Tube voltage correlated strongly with dose (R = 0.73, p<0.05). The ELCR values (0.37×10 to 0.96×10–3) exceeded the global reference of 0.29×10–3, indicating elevated lifetime oncogenic risk. Conclusion: This study highlights significant deficiencies in pediatric radiation safety, with environmental hotspots and inconsistent imaging protocols exceeding international benchmarks. Implementing standardized imaging techniques, enhanced shielding, and national pediatric reference levels is urgently needed to reduce long-term health risks in vulnerable populations.
INTRODUCTION
The rise in medical imaging across low- and middle-income countries has increased population exposure to ionizing radiation, often in settings where regulatory oversight and quality assurance are still evolving. Computed tomography and conventional radiography now contribute substantially to collective dose worldwide, with evidence showing a steady upward trend in medical exposure over the past few decades1-3. In Nigeria’s Niger Delta, this challenge is compounded by environmental stressors such as hydrocarbon pollution, industrial emissions, and geological sources of radon, all of which can elevate background ionizing radiation (BIR) in the built environment and potentially compromise radiation protection in hospitals3-5.
Several studies have documented spatial variations in environmental gamma radiation and associated cancer risks. Taskin et al.6 reported elevated natural gamma radioactivity and increased lifetime cancer risk in parts of Turkey, highlighting how local geology and land use can significantly modify background dose6. Similar findings have been observed in Pakistan, where indoor and outdoor gamma measurements sometimes exceeded global averages7. In Rivers State, Anekwe et al.4 showed that environmental gamma radiation levels in some industrial and residential zones are higher than UNSCEAR world averages, underscoring the need for continuous monitoring and risk assessment in this region.
Children represent the most vulnerable group in diagnostic radiology. They possess rapidly dividing tissues, smaller body mass, and a longer remaining lifespan, all of which increase the probability and lifetime impact of radiation-induced malignancies1,2,8,9. Brenner and Hall demonstrated that paediatric CT examinations contribute disproportionately to projected radiation-induced cancer risk, even when individual doses remain in the low-dose range1. For comparable exposures, the risk of solid cancers and leukemia is higher when irradiation occurs in childhood than in adulthood10,11. These concerns are amplified in settings where background radiation is already elevated.
International bodies such as the ICRP and UNSCEAR therefore recommend strict optimization of paediatric imaging, including the use of diagnostic reference levels (DRLs), protocol standardization, and regular quality assurance2,3. However, studies from Sudan, Brazil, Kenya, and Saudi Arabia have shown that paediatric entrance surface doses (ESDs) for common X-ray examinations can vary widely between centres, reflecting differences in equipment performance, exposure factors and operator training10-13. Suliman et al.10 for example, reported large inter-facility variations in paediatric ESDs in Sudan, similar inconsistencies in Kenyan and Saudi hospitals despite broadly comparable examinations13,14. These findings mirror the situation in many Nigerian facilities, where paediatric DRLs are absent, imaging charts are often adapted from adult protocols, and structured training in paediatric dose optimisation is limited6,8,9. This context underscores the importance of generating local data on both BIR and paediatric patient doses, as undertaken in the present study. This study provides a comprehensive assessment of environmental radiation levels and pediatric X-ray exposures at Rivers State University Teaching Hospital (RSUTH). The investigation aimed to (1) Quantify BIR around the hospital, (2) Measure pediatric diagnostic doses, (3) Compute associated cancer risks, and (4) Compare findings with international standards.
MATERIALS AND METHODS
Study area: The research was conducted at RSUTH, Port Harcourt, located in a high-industrial region of the Niger Delta. The hospital includes radiology units, laboratories, emergency wards, theaters, and administrative blocks. Data collection was carried out between June and August 2020, during the COVID-19 pandemic.
Study design: A cross-sectional design was employed, consisting of:
| • | Environmental BIR measurement across selected indoor and outdoor sites | |
| • | Pediatric dose assessment during diagnostic radiography |
Ethical approval for the study was obtained from the institutional review board, and written informed consent was secured from the parents or guardians of all participating children.
Background radiation measurement: A calibrated Digilert-200 Survey Meter (S.E. International) capable of detecting alpha, beta, gamma, and X-rays was used. Measurements were taken at 37 locations, including radiology, laboratories, waiting areas, and outdoor spaces. Each point was measured thrice, and the mean values were recorded.
The absorbed dose rate (nGy/hr), annual effective dose equivalent (AEDE), and excess lifetime cancer risk (ELCR) were calculated using UNSCEAR and ICRP conversion factors.
Where:
| OF | = | Occupancy factor | |
| DL | = | 70 years | |
| RF | = | 0.05 Sv | |
| D | = | Absorbed dose rate in air (nGy/hr) 8760 = Number of hours in a year | |
| 0.7 | = | Dose conversion coefficient from absorbed dose in air to effective dose (Sv/Gy) |
UNSCEAR (2000, 2008) Global Radiation Assessment Reports3,9.
Pediatric patient dosimetry: Thirty-nine pediatric patients aged 3-15 years undergoing common X-ray examinations (chest, skull, abdomen, neck, limb imaging) were enrolled. Optically Stimulated Luminescence (OSL) dosimeters were placed at the anatomical site of interest during each procedure.
Machine parameters (kVp, mAs, focus-to-skin distance) were recorded from the X-ray unit.
Entrance surface dose calculation: The Entrance Surface Dose (ESD) for pediatric radiographic examinations was computed using the internationally validated equation recommended by the International Atomic Energy Agency (IAEA) and widely used in diagnostic radiology dose surveys4,7,12.
Where:
| O/P | = | Tube output per mAs (mGy/mAs) at 80 kVp | |
| kV | = | Tube potential (kilovoltage peak) | |
| mAs | = | Tube current-time product (milliampere-seconds) | |
| FSD | = | Focus-to-skin distance (cm) | |
| BSF | = | Backscatter factor (dimensionless; typically, 1.30-1.35 for pediatric X-rays) |
IAEA Technical Reports & international pediatric radiography surveys4,7,12.
Statistical analysis: Data were evaluated using SPSS v25. Pearson correlation tested associations between kVp, mAs, FSD, and ESD. Findings were compared with IAEA pediatric DRLs. Descriptive statistics were expressed as means and standard deviations. Pearson’s correlation coefficient was used to evaluate the relationship between exposure parameters (kVp, mAs, FSD) and measured entrance surface dose (ESD). A significance level of p<0.05 was adopted as the criterion for statistical significance.
RESULTS
Background ionizing radiation (BIR): Background ionizing radiation measurements were obtained from 37 Locations within Rivers State University Teaching Hospital, comprising indoor and outdoor sites.
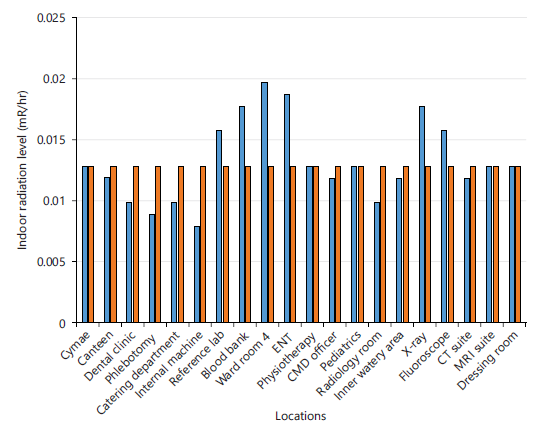
|
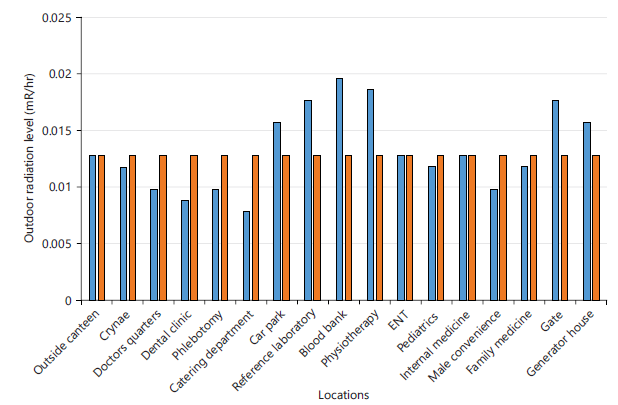
|
Figure 1 shows the measured indoor background ionizing radiation (BIR) levels compared with the ICRP reference limit of 0.013 mR/hr. Indoor radiation levels ranged from 0.008±0.002 to 0.020±0.002 mR/hr, with a mean value of 0.017±0.001 mR/hr. More than half (55%) of the indoor locations recorded values above the reference limit.
Figure 2 illustrates the outdoor BIR levels relative to the same ICRP reference standard. Outdoor radiation values ranged from 0.008±0.002 to 0.020±0.002 mR/hr, with a mean of 0.011±0.003 mR/hr, showing lower average exposure compared with indoor locations.
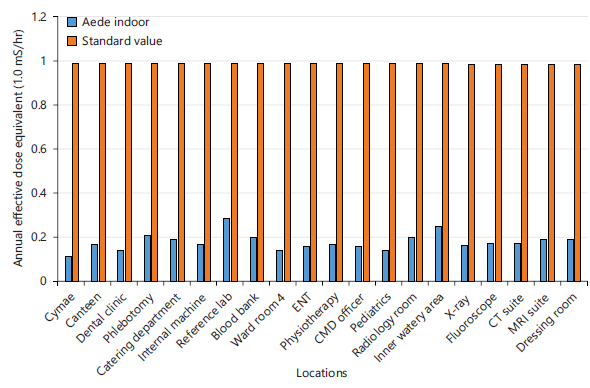
|
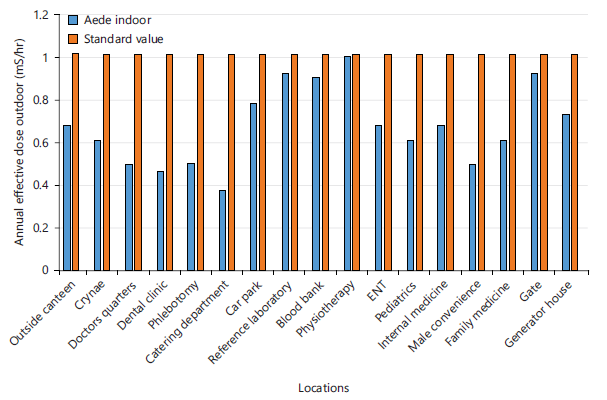
|
Figure 3 compares the indoor annual effective dose equivalent (AEDE) values at different hospital locations with the standard limit of 1.0 mSv/yr. Indoor AEDE values ranged from approximately 0.10 to 0.29 mSv/yr, with higher values observed in areas such as the Reference Laboratory and Inner Water Area. All indoor AEDE values were below the recommended limit.
Figure 4 displays the outdoor AEDE values, which ranged from 0.37 to 0.99 mSv/yr, indicating higher variability across outdoor locations, with all values remaining below the standard limit of 1.0 mSv/yr.
Pediatric X-ray dosimetry: A total of 39 pediatric patients aged 3-15 years were assessed during routine diagnostic radiography.
|
|
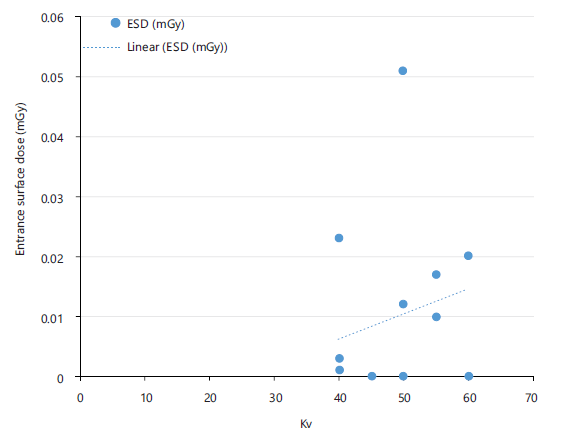
Figure 5 shows the relationship between entrance surface dose (ESD) and tube voltage (kVp) for chest X-ray examinations. ESD values ranged from 0.004 to 0.015 mGy, increasing with tube voltage in the range of 55-70 kVp.
|
|
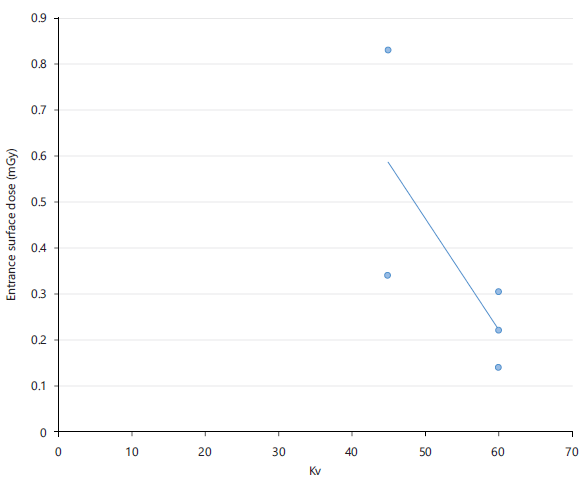
Figure 6 illustrates the ESD–kVp correlation for skull examinations, where ESD values ranged from 0.280 to 0.450 mGy at tube voltages between 65 and 85 kVp, indicating higher surface doses compared with chest examinations.
|
|
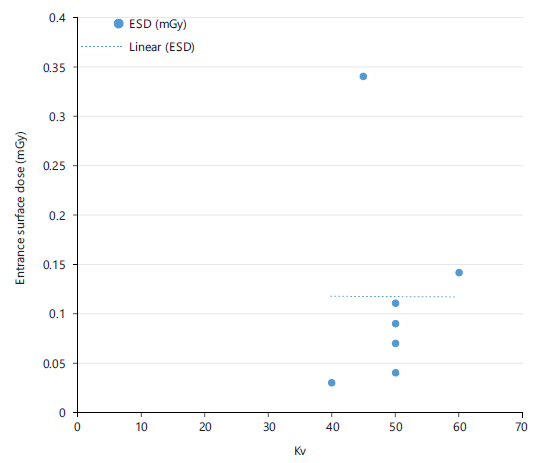
Figure 7 presents the correlation between ESD and kVp for neck examinations. The ESD values ranged from 0.050 to 0.090 mGy within a tube voltage range of 60-75 kVp.
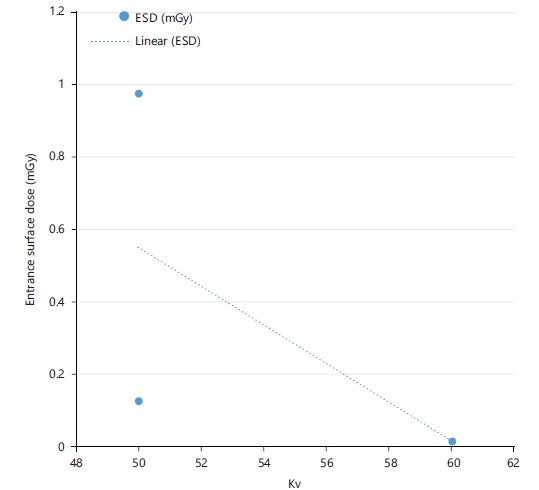
Figure 8 displays the ESD-kVp relationship for abdomen examinations. The linear trendline highlights this pattern, suggesting that optimizing tube voltage can effectively reduce patient radiation exposure. Notably, ESD varied between approximately 0.01 mGy and 0.97 mGy across the measured kV range (50-60 kV).
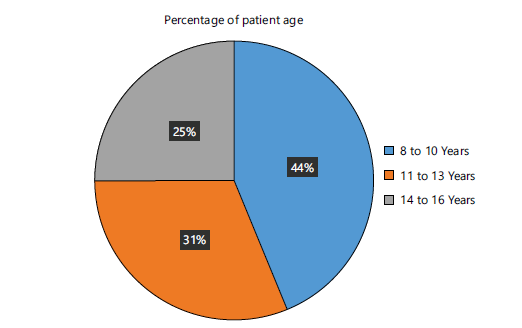
Figure 9 shows the percentage distribution of pediatric patients by age group. Patients aged 8-10 years constituted the largest group (44%), followed by 11-13 years (31%), 5-7 years (15%), and 14-15 years (10%).
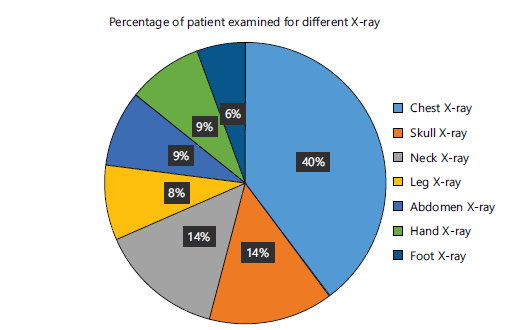
Figure 10 presents the percentage distribution of pediatric X-ray examinations performed. Chest radiography accounted for 40% of examinations, followed by skull (13%), neck (13%), abdomen (8%), hand (8%), leg (8%), and foot (5%).
A Pearson correlation analysis demonstrated a strong positive correlation between tube voltage and ESD (r = 0.73, p<0.05). No statistically significant correlation was observed between patient age or weight and measured ESD values.
DISCUSSION
The indoor and outdoor background ionizing radiation (BIR) levels measured at Rivers State University Teaching Hospital (RSUTH) fall within the broad range reported in environmental radiation surveys worldwide, but a notable proportion of indoor locations (55%) exceeded the ICRP-recommended reference level of 0.013 mR/hr. This pattern is consistent with earlier work in the Niger Delta, where Taskin et al.6 reported elevated gamma dose rates in parts of Rivers State, particularly in areas impacted by industrial activities and hydrocarbon operations6. Similar trends have been documented elsewhere: Taskin et al.6 found that natural gamma radioactivity in parts of Turkey led to dose rates and excess lifetime cancer risks above global averages, while indoor and outdoor gamma levels in Pakistan that, in some locations, also surpassed UNSCEAR world mean values.
The present study’s AEDE values (0.11-0.29 mSv/yr) are lower than the 1 mSv/yr public dose limit recommended by ICRP2,3,11, aligning with observations from several environmental surveys in low- and middle-income settings where chronic exposure remains within regulatory thresholds, even when localized hotspots exist5,6,15. However, the computed ELCR values (0.37×10–3-0.96×10–3) are higher than the commonly referenced global nominal risk benchmark of 0.29×10–3 5,11, reinforcing concerns raised by prior authors that seemingly modest increases in background dose can translate into non-negligible long-term cancer risk when applied to large populations3,5.
Overall, the BIR profile at RSUTH corroborates earlier regional findings that the Niger Delta is a radiologically sensitive environment due to hydrocarbon pollution, gas flaring, and NORM mobilization6,16. By combining environmental monitoring with clinical dosimetry, this study extends those observations into the hospital setting and provides a more patient-centered view of risk.
The pediatric entrance surface dose (ESD) values obtained in this study for chest, skull, and abdomen radiography fall within or below the ranges reported in comparable international surveys. Pediatric ESD values for chest X-rays in Sudan were generally higher than those observed at RSUTH, with substantial inter-hospital variability reflecting differences in equipment performance and technique. de Azevedo et al.11 similarly documented wide ESD ranges at a Brazilian pediatric hospital, emphasizing that protocol optimization can significantly reduce dose without compromising image quality.
In Kenya, pediatric ESDs for chest and skull examinations were often above suggested DRLs, and highlighted the need for local reference values and systematic dose audits13. pediatric ESDs for common examinations in Saudi Arabia that were comparable to or slightly higher than those found in this study, again reinforcing the global pattern of variability in pediatric dosing practice14. The lower ESDs observed for chest radiography at RSUTH (mean 0.009 mGy) relative to many of these studies4,12-14 suggest that, for some projections, exposures at this facility are reasonably optimized. However, the relatively higher doses for skull and abdomen examinations (0.368 and 0.371 mGy, respectively) and the wide range in ESD values point to non-uniform technique and potential opportunities for further optimization.
The strong positive correlation between tube voltage (kVp) and ESD (r = 0.73, p<0.05) aligns with the physics of X-ray generation and with findings showed that patient doses in diagnostic radiology are highly sensitive to kVp, mAs, and focus-to-skin distance7. Protocol revisions targeting these parameters could achieve meaningful dose reductions in a pediatric X-ray department8. Systematic adjustment and standardization of exposure parameters are central to managing patient doses across different facilities9.
The present study confirms, within a Nigerian context, that variations in technical factors remain a primary driver of pediatric dose variability, underscoring international calls for protocol-based control of exposure settings2,4,7-9,12-14.
The ELCR values derived from the combined environmental and diagnostic exposures in this study are particularly relevant in light of pediatric radiosensitivity. Children exposed to diagnostic X-rays and CT scans carry a proportionally higher lifetime risk of radiation-induced cancer compared with adults at similar dose levels1. biological mechanisms, noting that rapidly proliferating tissues and longer post-exposure life expectancy amplify the impact of even low- to moderate-dose exposures10,11.
Although the individual effective doses per examination observed in this study are low and broadly consistent with ICRP and UNSCEAR assumptions for diagnostic procedures2,3,11, the elevated ELCR estimates for the hospital environment, combined with repeated imaging in some patients, suggest that cumulative risk cannot be ignored. UNSCEAR and ICRP both stress that stochastic risks add up over a lifetime and that children, in particular, benefit most from aggressive dose optimization and exposure justification2,3,11. In this way, the present findings align with international risk frameworks while highlighting the added burden posed by elevated background radiation in the Niger Delta environment3,6,16.
Collectively, the BIR, ESD, and ELCR findings from RSUTH echo patterns reported in other low- and middle-income countries, where infrastructure, training, and regulation often lag behind technological adoption4,7-9,12-14. The lack of nationally defined Pediatric Diagnostic Reference Levels (P-DRLs) in Nigeria mirrors the situation previously described in Sudan, Kenya, and parts of the Middle East, where local data had to be generated before optimization efforts could be meaningfully implemented4,13,14.
The present study contributes such baseline data for a major Nigerian teaching hospital and illustrates how environmental and clinical measurements can be integrated into a unified radioprotection strategy. It also reinforces recommendations that patient dose management should be viewed not as a one-time exercise, but as a continuous process involving protocol review, staff training, equipment quality control, and feedback based on measured doses7-9. In the Niger Delta context, this continuous improvement model must also account for elevated environmental radiation and potentially greater cumulative risk.
Several limitations should be acknowledged. First, the sample size for pediatric dosimetry (39 patients) is relatively small and unevenly distributed across examination types, which restricts the ability to establish robust local DRLs or to perform detailed subgroup analyses by age, weight, or clinical indication. Second, the study relied on OSL dosimeters placed at the entrance surface and did not compute organ-specific or effective doses using patient-specific Monte Carlo simulations or computational phantoms; thus, risk estimates are based on standard conversion factors rather than individualized dose reconstructions. Third, BIR measurements were conducted during a single campaign and may not fully capture temporal variations related to occupancy patterns, equipment use, or seasonal environmental changes.
In addition, the analysis did not systematically evaluate image quality in parallel with dose, which is a key component of true optimization; hence, some low ESD values might be associated with suboptimal diagnostic quality in practice, although this was not formally assessed. Finally, the study was confined to a single teaching hospital, limiting the generalizability of findings to other Nigerian or Sub-Saharan facilities with different equipment, workloads, or environmental conditions.
Future research should therefore focus on multi-center studies involving larger pediatric cohorts and a wider range of examination types, with harmonized data collection across Nigeria and the wider region. Integration of Monte Carlo-based organ dose estimation and pediatric computational phantoms would enhance the accuracy of risk assessment. Longitudinal BIR monitoring, including radon measurements and seasonal variation analysis, would clarify the stability and determinants of environmental exposure in hospital environments. There is also a need to link dose measurements with image quality assessment and clinical outcomes to develop evidence-based pediatric DRLs that reflect both safety and diagnostic performance.
Finally, targeted intervention studies such as protocol optimization, staff training, and equipment upgrades should be implemented and evaluated using pre- and post-intervention dosimetric audits. Such work would support the development of a national framework for pediatric radiation protection, which is particularly crucial in environmentally burdened regions like the Niger Delta.
CONCLUSION
The study revealed elevated background radiation, variable pediatric X-ray exposures, and above-threshold cancer risk indicators at RSUTH. Although measured ESDs were generally within acceptable limits, environmental hotspots and inconsistent imaging protocols pose significant long-term risks. To enhance pediatric radiation safety in Nigeria, establishing National Pediatric Diagnostic Reference Levels (P-DRLs), implementing routine quality assurance with equipment calibration, strengthening shielding in high-exposure areas, training radiographers in dose optimization, and developing a national pediatric dose registry are essential measures. These actions will safeguard vulnerable populations and ensure compliance with international safety standards.
SIGNIFICANCE STATEMENT
This study discovered elevated background ionizing radiation levels and excessive pediatric X-ray doses that can be beneficial for developing targeted radiation protection strategies in hospitals. It emphasizes the correlation between tube voltage and dose, highlighting environmental hotspots and procedural inconsistencies. This study will help the researchers to uncover the critical areas of pediatric radiation safety that many researchers were not able to explore. Thus, a new theory on optimizing radiation protection for vulnerable populations may be arrived at.
ACKNOWLEDGMENT
The authors appreciate the staff and management of Rivers State University Teaching Hospital for their cooperation throughout this study.
REFERENCES
- Brenner, D.J. and E.J. Hall, 2007. Computed tomography-an increasing source of radiation exposure. N. Engl. J. Med., 357: 2277-2284.
- Valentin, J., 2007. The 2007 Recommendations of the International Commission on Radiological Protection. ICRP Publication 103, Essen, Germany, ISBN: 978-0-7020-3048-2, Pages: 332.
- UNSCEAR, 2000. Sources and Effects of Ionizing Radiation. United Nations Publications, New York, USA, ISBN:92-1-142238-8, Pages: 654.
- Anekwe, U.L., G.O. Avwiri and E.O. Agbalagba, 2013. Assessment of gamma-radiation levels in selected oil spilled areas in Rivers State, Nigeria. Energy Sci. Technol., 5: 33-37.
- Agbalagba, E.O., G.O. Avwiri and Y.E. Chadumoren, 2013. Radiological impact of oil and gas activities in selected oil fields in production land area of Delta State, Nigeria. J. Appl. Sci. Environ. Manage., 17: 279-288.
- Taskin, H., M. Karavus, P. Ay, A. Topuzoglu, S. Hidiroglu and G. Karahan, 2009. Radionuclide concentrations in soil and lifetime cancer risk due to gamma radioactivity in Kirklareli, Turkey. J. Environ. Radioact., 100: 49-53.
- Zeb, J., M. Wasim, M. Awais, Asad Ullah, T. Iqbal and S. Akhtar, 2020. Evaluation of indoor/outdoor gamma exposure rates and excess life time cancer risk in different cities of Pakistan. Radiat. Prot. Dosim., 190: 355-363.
- Pearce, M.S., J.A. Salotti, M.P. Little, K. McHugh and C. Lee et al., 2012. Radiation exposure from CT scans in childhood and subsequent risk of leukaemia and brain tumours: A retrospective cohort study. Lancet, 380: 499-505.
- UNSCEAR, 2008. Sources and Effects of Ionizing Radiation. United Nations Publications, New York, USA, ISBN: 978-92-1-142274-0.
- Suliman, I.I., N. Abbas and F.I. Habbani, 2007. Entrance surface doses to patients undergoing selected diagnostic X-ray examinations in Sudan. Radiat. Prot. Dosim., 123: 209-214.
- de Azevedo, A.C.P., A.O. Osibote and M.C.B. Boechat, 2006. Survey of doses and frequency of X-ray examinations on children at the intensive care unit of a large reference pediatric hospital. Appl. Radiat. Isot., 64: 1637-1642.
- Horst, K.K., Z. Zhou, N.C. Hull, P.G. Thacker and B.A. Kassmeyer et al., 2025. Radiation dose reduction in pediatric computed tomography (CT) using deep convolutional neural network denoising. Clin. Radiol., 80.
- Johnson, C., T. Martin-Carreras and D. Rabinowitz, 2014. Pediatric Interventional Radiology and dose-reduction techniques. Semin. Ultrasound CT MRI, 35: 409-414.
- Uffmann, M. and C. Schaefer-Prokop, 2009. Digital radiography: The balance between image quality and required radiation dose. Eur. J. Radiol., 72: 202-208.
- Mettler, F.A., W. Huda, T.T. Yoshizumi and M. Mahesh, 2008. Effective doses in radiology and diagnostic nuclear medicine: A catalog. Radiology, 248: 254-263.
- Kim, Y.H., J.H. Choi, C.K. Kim, J.M. Kim and S.S. Kim et al., 2007. Patient dose measurements in diagnostic radiology procedures in Korea. Radiat. Prot. Dosim., 123: 540-545.
How to Cite this paper?
APA-7 Style
Chidinma,
O.L., Chad-Umoren,
Y.E., Frank,
A.O. (2026). Ionizing Radiation and Pediatric X-ray Assessment in Rivers State, Nigeria. Asian Journal of Emerging Research, 8(1), 60-71. https://doi.org/10.21124/ajer.2026.60.71
ACS Style
Chidinma,
O.L.; Chad-Umoren,
Y.E.; Frank,
A.O. Ionizing Radiation and Pediatric X-ray Assessment in Rivers State, Nigeria. Asian J. Emerg. Res 2026, 8, 60-71. https://doi.org/10.21124/ajer.2026.60.71
AMA Style
Chidinma
OL, Chad-Umoren
YE, Frank
AO. Ionizing Radiation and Pediatric X-ray Assessment in Rivers State, Nigeria. Asian Journal of Emerging Research. 2026; 8(1): 60-71. https://doi.org/10.21124/ajer.2026.60.71
Chicago/Turabian Style
Chidinma, Olinya, Lilian, Yehuwdah E. Chad-Umoren, and Adizua Okechukwu Frank.
2026. "Ionizing Radiation and Pediatric X-ray Assessment in Rivers State, Nigeria" Asian Journal of Emerging Research 8, no. 1: 60-71. https://doi.org/10.21124/ajer.2026.60.71

This work is licensed under a Creative Commons Attribution 4.0 International License.



