Analysis of Bioactive Compounds Present in the Leaf Extracts of Senna alata, Dennettia tripetalla and Delonix regia
| Received 24 Oct, 2020 |
Accepted 02 Jan, 2021 |
Published 07 Jun, 2021 |
Background and Objectives: Tropical shrubs have been in use as medicinal and ornamental plants by traditional herbalists in Southern Nigeria in the management and treatment of various illnesses. Materials and Methods: The Gas Chromatography-Mass Spectrometry (GC-MS) of the dichloromethane extracts of Senna alata, Dennettia tripetalla and Delonix regia leaves were carried out to analyze the bioactive compounds present in the leaves of these plants. Ten grams (10 g) of each of the powdered samples were extracted with 20 mls of dichloromethane, concentrated on a steam bath to about 5ml, purified by passing through a pasture pipette packed with silica gel and anhydrous sodium sulphate on a membrane and air-dried to about 2ml for gas chromatographic analysis. The dichloromethane concentrate of the various extracts was diluted with 98% hexane and 1μ/l of each diluted sample was automatically injected into the Gas chromatographic Model: 7890A (GC) interfaced with Mass Selective Detector model: 5975C (MSD). Identification of bioactive compounds present in the different extracts was based on GC retention time on HP- 5 column and matching of the spectra with computer software using the Chem-software attached to the MS library. Detection of compounds present in each leaf sample was confirmed using the database of the National Institute of Standards and Technology (NIST). Results: The results showed that the leaf extract of S. alata, had the highest peaks observed for Vitamin E (38.338%) and n-Hexadecanoic acid (21.695%), at retention times of 33.419 and 20 minutes respectively. D. regia leaf extract had the highest peaks observed for Dodecanoic acid, 1, 2, 3-propanetriyl ester (37.228%) and Phytol (34.681%) at retention times of 33.664 and 22.535 minutes respectively. Also, the results from D. tripetalla extracts showed that the highest peaks were observed for Supraene (27.91%) followed by 5, 9, 13-pentadecatriene-2-one/6, 10, 14-trimethyl/(E, E) (27.778%) at retention times of 30.897 and 19.441 minutes respectively. Conclusions: The analysis of the dichloromethane leaf extracts of the three plants showed that they contain bioactive compounds which can be explored for nutraceutical and medicinal uses.
INTRODUCTION
The use of extracts, from medicinal plants, as therapeutic agents has been an important area in traditional and natural product research1. These plants have a wild range of nutritional and pharmacological activities. Nutritionally, most plants contain biological molecules such as amino acids, proteins, carbohydrates, fats, and oils. Their pharmacological activities have been channeled for use including as anti-spasmodic, anti-hypertensive, anti-microbial, anti-fungi and laxative2. These plants contain several chemical compounds essential for the metabolic functioning of our body systems. Some of these plants provide us food, nutritional values, and health benefits while some provide us with no nutrients and could even be toxic to human health. As a result, identifying these beneficial plants are highly imperative as a guide for gaining all the benefits they offer3. Plants can be a source of chemical compounds of biological and pharmacological importance, and will continuously be important for the screening of novel naturally occurring chemical entities4. An important part of the investigation of the plant is the identification of the bioactive secondary metabolites they possess leading to further biological and pharmacological studies5-7. Some traditional plants used by the locals, in Southern Nigeria, to treat various illnesses include Senna alata, Denettia tripetalla, and Delonix regia.
Delonix regia (Hook.) Raf. is a specie in the genus Delonix (family Leguminosae)8. It isfound in tropical countries and is referred to as the royal Poinciana flamboyant or flame tree. The different parts of the tree are used in traditional medicine.The amount and chemical composition of the extracts of the different parts is dependent on tree species, tree partage, season, and location of the tree9. The extracts of D. regia have been reported to consist of mixtures of various components such as anthocyanin, carotenoids, flavonol, and phenolic acid from its flowers10. Various types of non-polar compounds, including free fatty acids such as myristic, palmitic, stearic, Oleic, and linoleic, have been found in the seed oil of D. regia11. The bark and flowers of D. regia have been reported to have broad-spectrum antibacterial, anti-fungal, and anti-inflammatory properties. The antibacterial properties of D. regia bark extract might be due to the presence of flavonoids, alkaloids and phenolic compounds12. It has previously been reported that high concentrations of polyphenol compounds including anthocyanins, flavones, and phenolic acid, found in wood and bark of D. regia are the most bioactive natural compounds given their antioxidant and antibacterial properties13. Aqueous extracts of D. regia seeds have been reported to exhibited antidiabetic activity, lowered pancreatic amylase activities and showed antioxidant properties in treated diabetic rats, and also exhibited greater effectiveness in resolving adverse effects in the pancreas and heart of high-fat diet streptozotocin-induced diabetic rats14.Phytochemical investigation revealed that the extracts of the stem bark contained – sitosterol, Lupeol, Cpi lupeol stigmasterol, and -methoxybenzal dehyde saponins, alkaloids, carotene, hydrocarbons phytoboxins and flavonoids13,15, gallic acid, phenolic acids such as sorbic sinapic, -coumaric, M-coumaric, ferulic, caffeic, 3-hydroxybenzoic, 4-hydroxycinnamic, and 4-hydroxybezoic acids16. The leaves are reported to have antibacterial and antimalarial properties17.
Senna alata (L) Roxb could serve as both a medicinal and an ornamental plant18. They grow well in full sun in a wide range of soils that retain moisture adequately. Studies have reported the use of S. alata (L) Roxb leaves in treating abdominal pain, constipation, and liver abnormalities19, eczema, skin inflammation, rashes on the skin, athlete’s foot, and ringworm (from where the name ‘ringworm shrub’ was derived)20. The leaf of S. alata (L) Roxb has been shown to possess significant hypolipidemic properties and thus, may be used in the management and treatment of hyperlipidemia21.
Dennettia tripetalla, also known as pepper fruit is widely grown in the rain forest zones of Nigeria and some parts of West Africa. The plant usually produces fruit between March and May. The fruits and seeds are edible and are consumed because of their spicy nature as well as their nutritional and health benefits22. D. tripetalla has a lot of medicinal potentials that are starting to be verified scientifically. The seeds are effective in reducing the intraocular pressure of normotensive emmetropic humans. Research23-24, showed that D. tripetalla can reduce the plasma glucose level in drug-induced hyperglycemic rats to levels comparable with that of normal rats. The essential oil of D. tripetalla fruits has been found to possess analgesic effects as compared to that induced by morphine, aspirin and indomethacin. This oil also exerted an anti-inflammatory effect on rodents with edema to levels comparable with that of dexamethasone25. Scientific evidence abounds that this plant has potential for use in the field of biotechnology in areas such as meat preservation26, pest control27, food supplementation and spicing28. In this study, the leaves of S. alata, D. tripetalla, and D. regia were investigated to observe the presence of bioactive compounds that can be beneficial to human health.
MATERIALS AND METHODS
Sample collection and identification: The leaves of S. alata, D. tripetalla, and D. regia were collected, authenticated and deposited at different herbariums as respectively described by Onyegeme-Okerenta and Anacletus29, Omeodu et al.30, Onyegeme-Okerenta et al.31. They were air-dried at room temperature (29±1°C) for 3 weeks and then pulverized with the aid of Marlex Excellent grinder (Mumbai, India). The ground samples were then passed through a sieve of 0.5 mm pore size to obtain a fine uniform powder. The powdered samples were kept in an airtight container until required.
Sample Extraction: Ten grams (10 g) of each of the powdered samples were weighed into a well-stoppered bottle and 20 mls of the dichloromethane was added. The mixtures were vigorously agitated and were left to stand for 5 days. Each crude extract was collected by filtering into a quartz beaker and the process was repeatedly carried out for two more consecutive times. The combined aliquots of each extract collected were concentrated on a steam bath to about 5 ml. This was purified by passing through a pasture pipette packed with silica gel and anhydrous sodium sulphate on a membrane and air-dried to about 2 ml for gas chromatographic analysis.
GC-MS Analysis of the leaf extractsof S. alata, D. tripetalla, and D. regia: The dichloromethane concentrate of the various extracts was diluted with 98% hexane and 1 μ/l of each diluted sample was automatically injected into the Gas chromatographic Model: 7890A (GC) interfaced with Mass Selective Detector model: 5975C (MSD) for Gas Chromatography/Mass Spectrometry analysis and characterization of the various bioactive compounds present32.
Identification of chemical constituents: Bioactive compounds present in the different extracts were identified based on GC retention time on HP- 5 column and matching of the spectra with computer software using the Chem-software attached to the MS library. Detection of compounds present in each leaf sample was confirmed using the database of the National Institute of Standards and Technology (NIST) which houses more than 62,000 patterns. The spectrum of the unidentified component was compared with the spectrum of the identified components stored in the NIST library. The name, molecular weight, structure of the components in the test material were then ascertained32.
RESULTS AND DISCUSSION
The bioactive compounds with their molecular formulae, molecular weight, Retention Time (RT), Peak area (%), are shown in Tables 1-3 respectively, while the Chromatogram of bioactive compounds in dichloromethane extracts and structure of the bioactive components identified in the dichloromethane extracts of S. alata, D. tripetalla, and D. regia leaves analyzed by GC-MS are presented in Fig. 1-24.
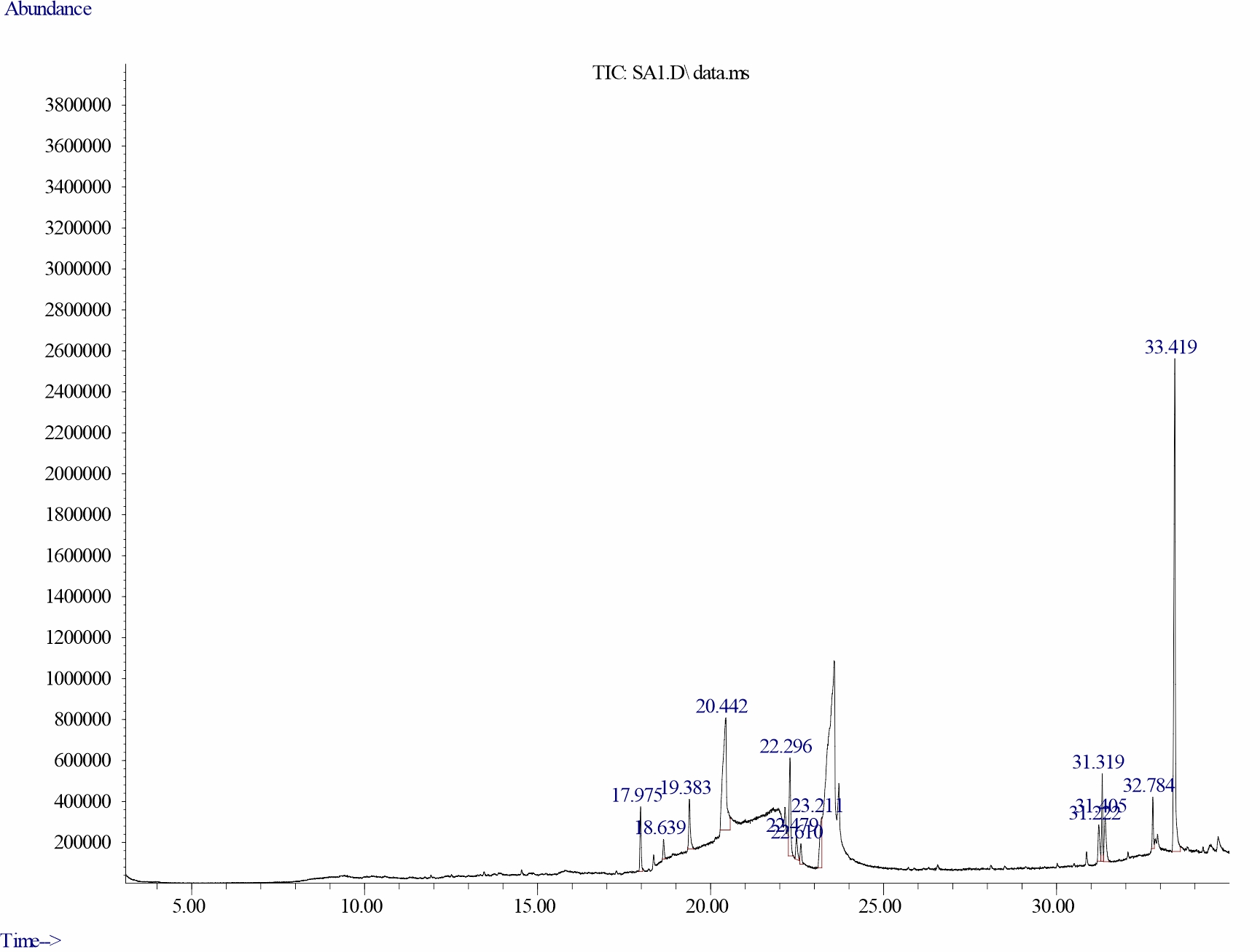
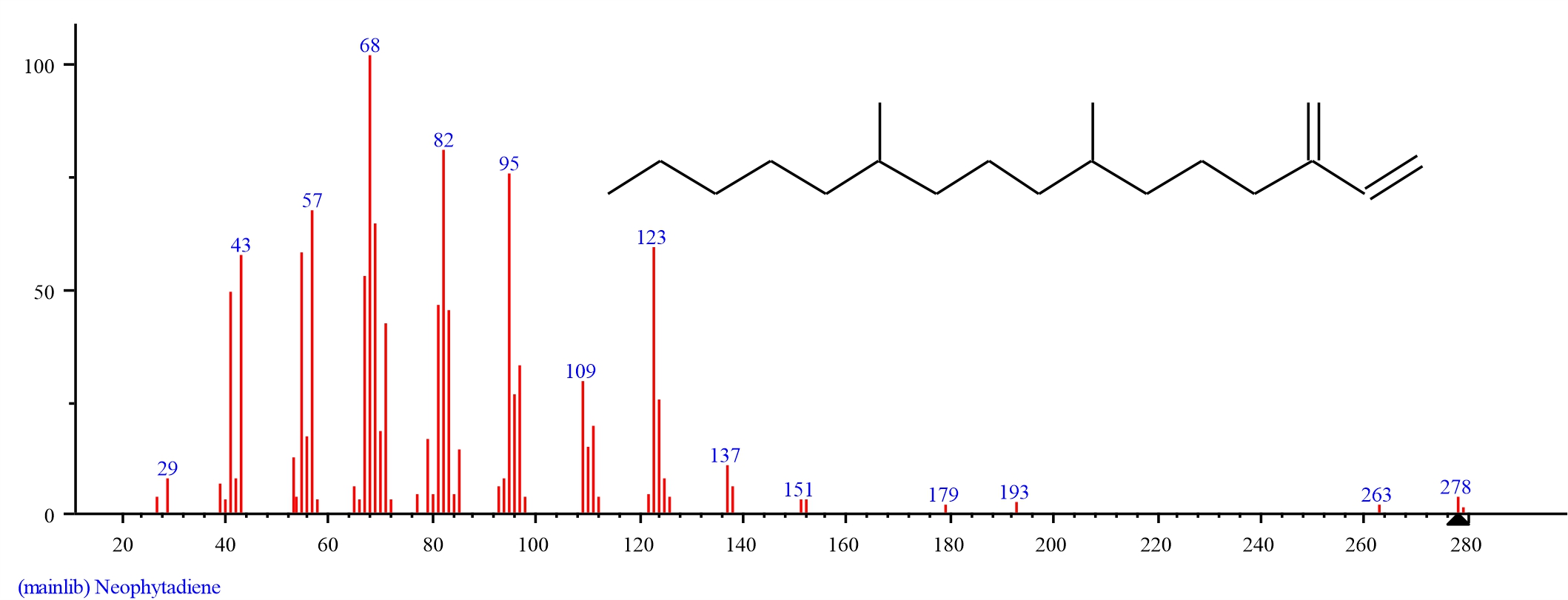
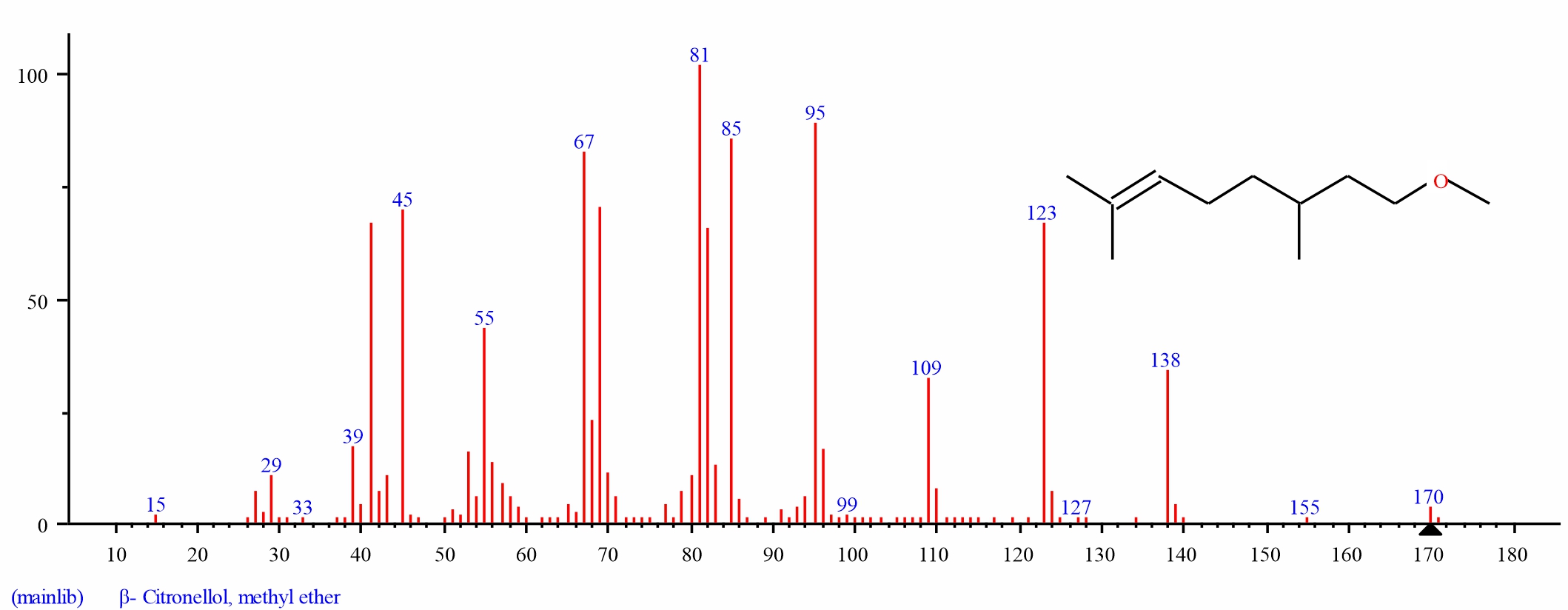
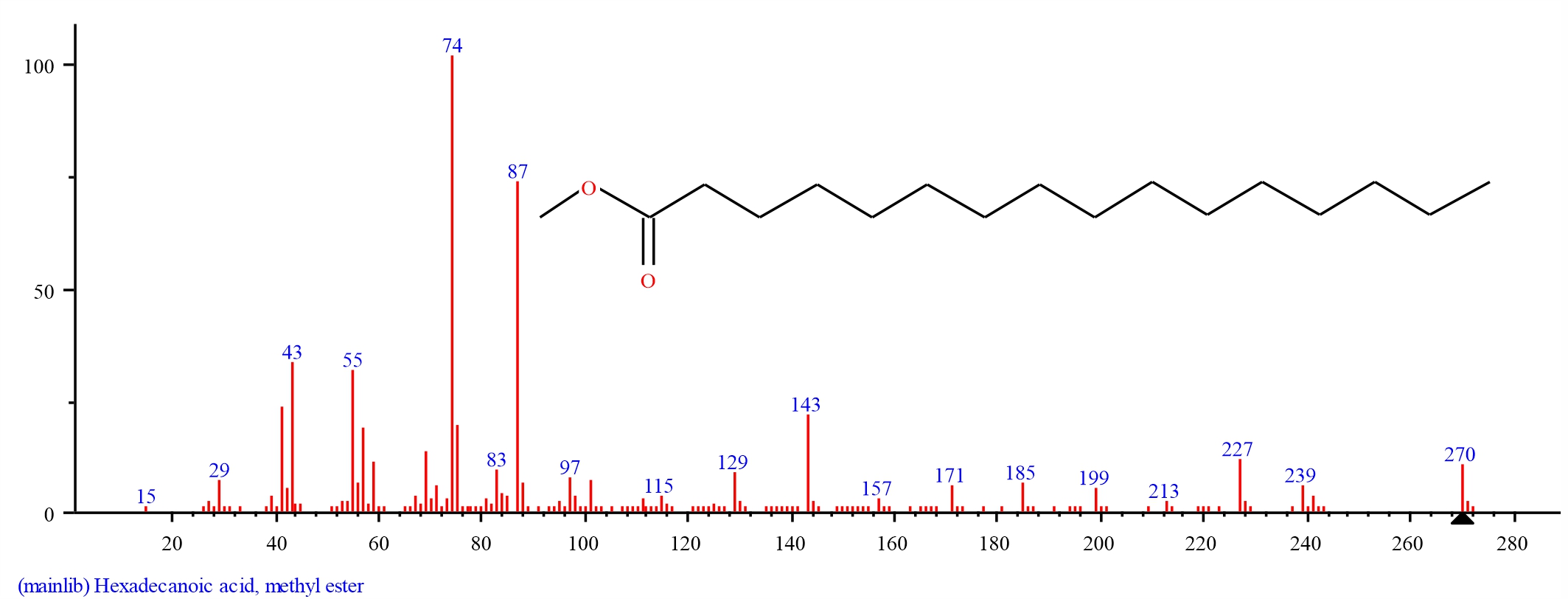
Gas chromatography-mass spectrometry analysis of dichloromethane extracts of S. alata, leaves: Gas chromatography-mass spectrometry (GC–MS) analysis of dichloromethane leaf extract of S. alatarevealed the presence of thirteen major bioactive compounds, their retention time and their final peak value (Table 1).The structure of each bio active compound was also elucidated. They are Neophytadiene (3.934), beta-Citronellol, methyl ether (1.187), Hexadecanoic acid, methyl ester (4.039), n-Hexadecanoic acid (21.695), 9, 12, 15-Octadecatrienoic acid, methyl ester, (Z, Z, Z)-(7.634), Phytol (1.877), Methyl stearate (1.478), 9, 12-Octadecadienoic acid (Z, Z)-( 5.289). alpha.-Tocospiro A (3.021), Nonacosane (4.487), alpha.-Tocospiro B (4.317), Tricosane (2.704) and Vitamin E (38.338). From the results of the GC–MS spectra, Vitamin E, n-Hexadecanoic acid,9, 12, 15-Octadecatrienoic acid methyl ester, (Z, Z, Z), 9, 12-Octadecadienoic acid (Z, Z), Nonacosane, alpha.-Tocospiro B, Hexadecanoic acid methyl ester, Neophytadiene, alpha.-Tocospiro A was the most abundant in occurrence whileTricosane, Phytol, Methyl stearate, beta-Citronellol methyl ether were the least(Fig. 1-14).
 |
||||||||||||||||||||||||||||||||||||||||||||||||||
Fig. 1: Chromatogram of bioactive compounds in dichloromethane extract of S. alata showing Vitamin E having a retention time of 33.419 minutes and final peak value of 38.338% |
|
|||||||
| S/N | Compound | Retention Time (min) | Peak Area (%) | Molecular formula | Molecular weight | ||
| 1 | Neophytadiene | 17.975 | 3.934 | C20H38 | 278.5157 | ||
| 2 | beta-Citronellol, methyl ether | 18.639 | 1.187 | C11H22O | 170.29 | ||
| 3 | Hexadecanoic acid, methyl ester | 19.383 | 4.039 | C17H34O2 | 270.4507 | ||
| 4 | n-Hexadecanoic acid | 20.442 | 21.695 | C16H32O2 | 256.4241 | ||
| 5 | 9,12,15-Octadecatrienoic acid, methyl ester, (Z,Z,Z)- | 22.296 | 7.634 | C19H32O2 | 292.4562 | ||
| 6 | Phytol | 22.479 | 1.877 | C20H40O | 296.539 | ||
| 7 | Methyl stearate | 22.610 | 1.478 | C19H38O2 | 298.511 | ||
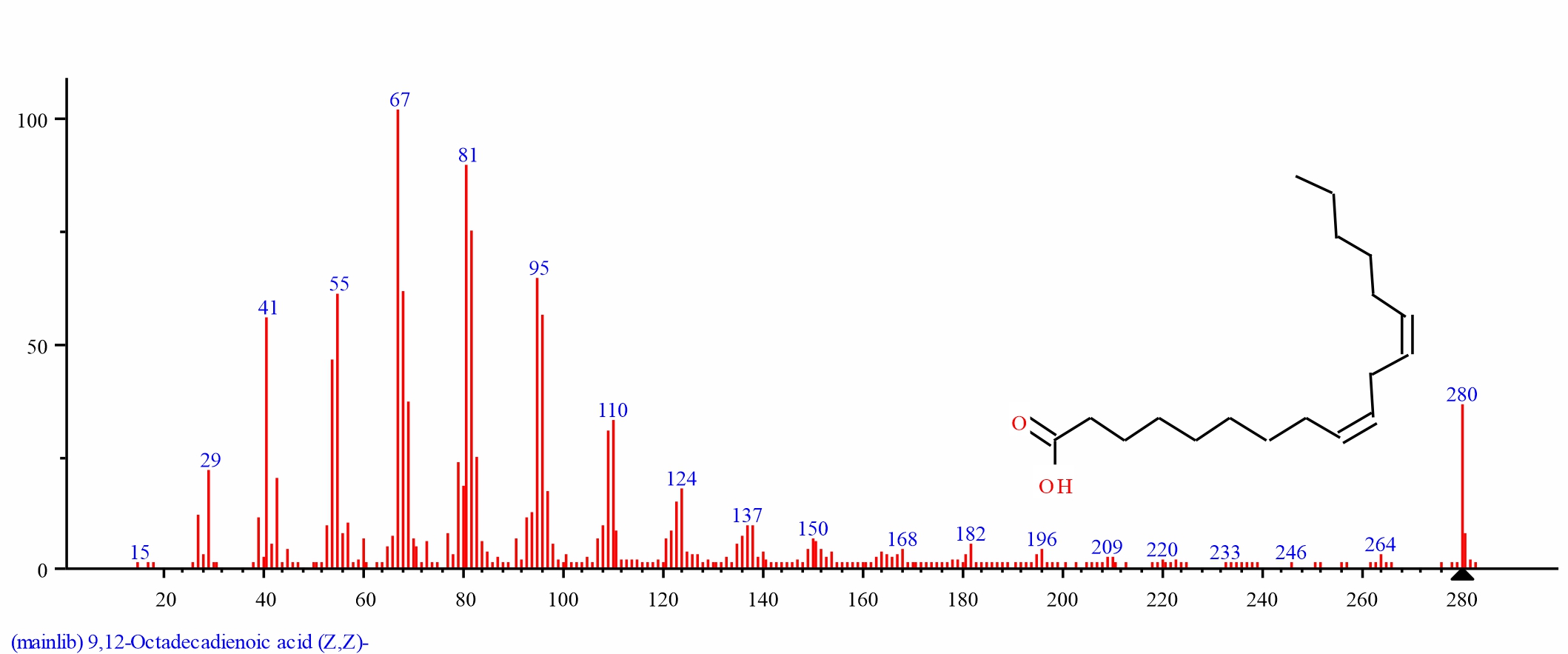
| 8 | 9,12-Octadecadienoic acid (Z,Z)- | 23.211 | 5.289 | C18H32O2 | 280.4455 | ||
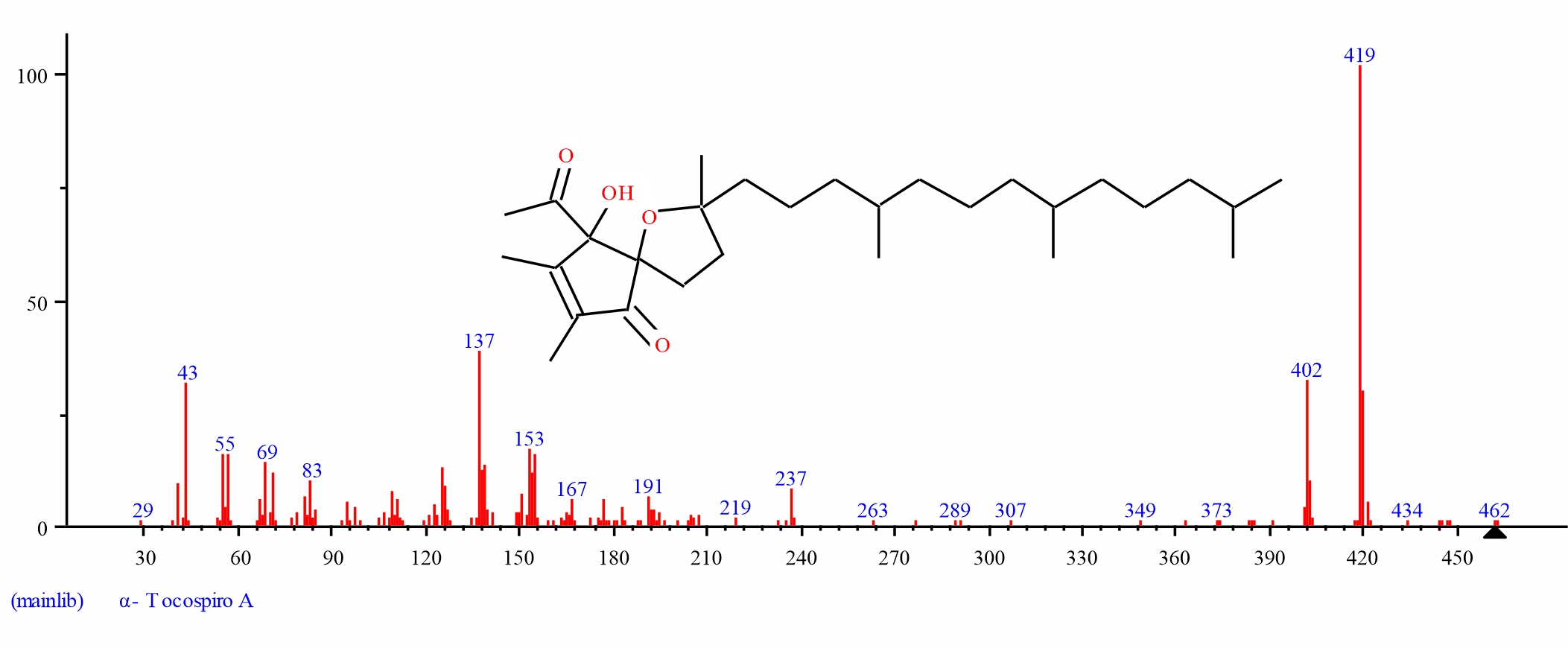
| 9 | alpha.-Tocospiro A | 31.222 | 3.021 | C29H50O4 | 462.715 | ||
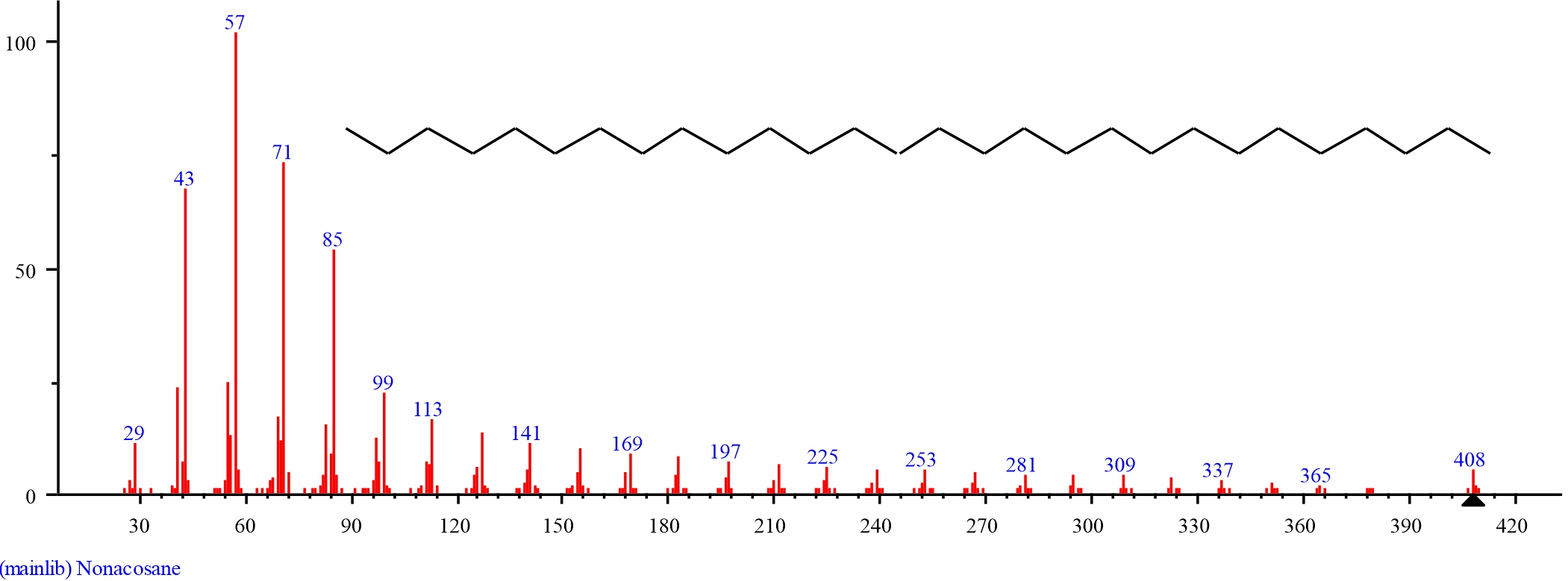
| 10 | Nonacosane | 31.319 | 4.487 | C29H60 | 408.7867 | ||
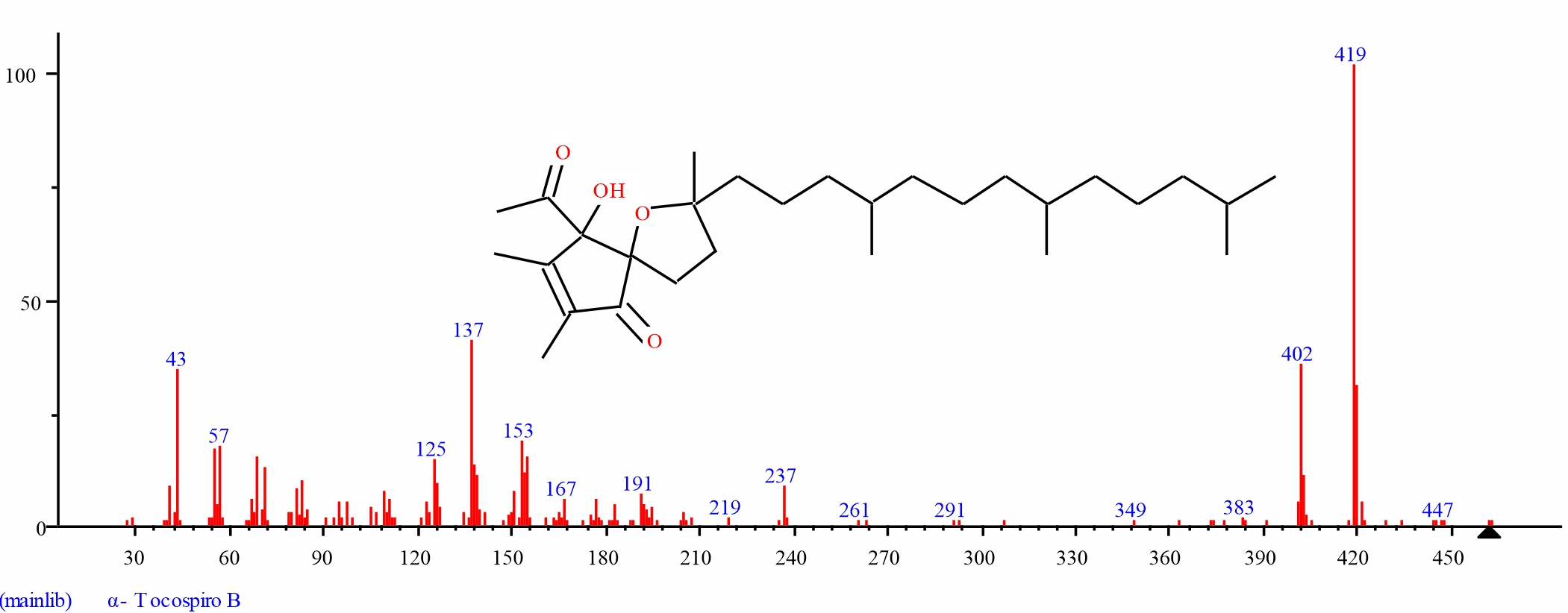
| 11 | alpha.-Tocospiro B | 31.405 | 4.317 | C29H50O4 | 462.715 | ||
| 12 | Tricosane | 32.784 | 2.704 | C23H48 | 324.637 | ||
| 13 | Vitamin E | 33.419 | 38.338 | C29H50O2 | 430.717 | ||
 |
||||||||||||||||||||||||||||||||||||||||||||||||||
Fig. 2: GC-MS spectra of Neophytadiene structure (3.934%, RT 17.975) from S.alata leaves |
 |
||||||||||||||||||||||||||||||||||||||||||||||||||
Fig. 3: GC-MS spectra of beta-Citronellol, methyl ether structure (1.187%, RT 18.639) from S. alata leaves |
 |
||||||||||||||||||||||||||||||||||||||||||||||||||
Fig. 4: GC-MS spectra of Hexadecanoic acid, methyl ester structure (4.039%, RT 19.383) present in S. alata leaves |
 |
||||||||||||||||||||||||||||||||||||||||||||||||||
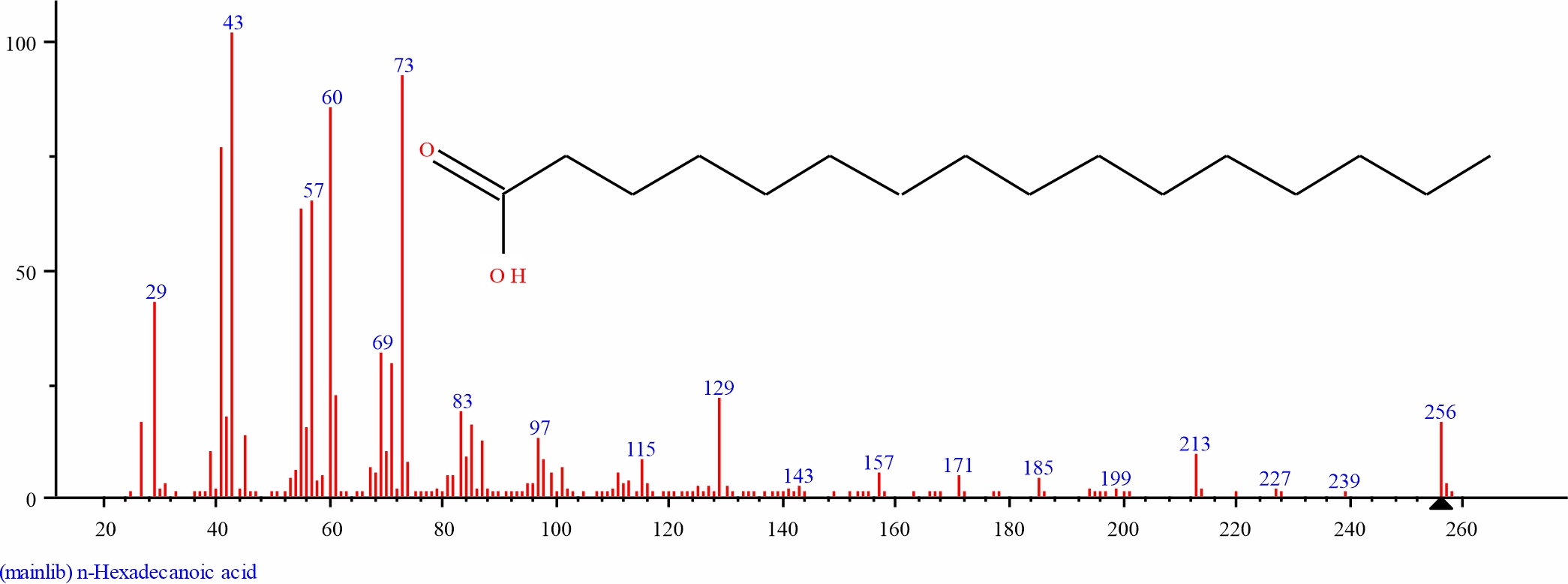
Fig. 5: GC-MS spectra of n-Hexadecanoic acid structure (21.695%, RT 20.442) from S. alata leaves |
 |
||||||||||||||||||||||||||||||||||||||||||||||||||
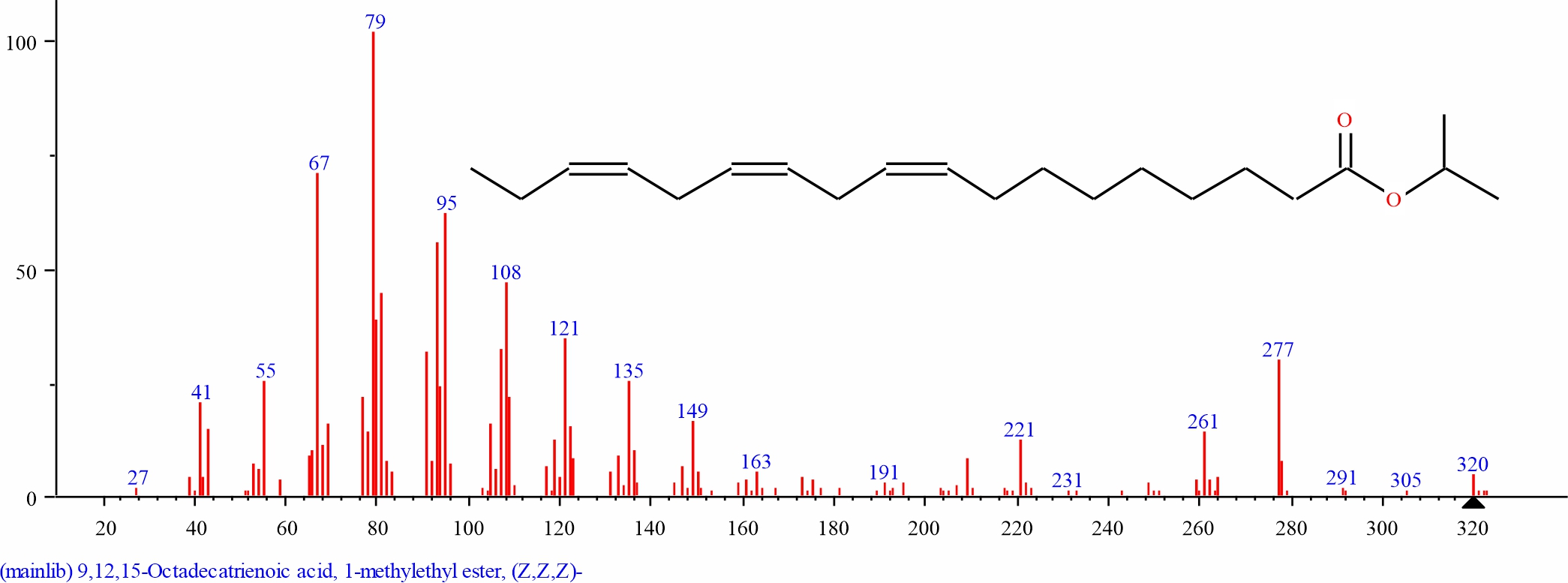
Fig. 6: GC-MS spectra of 9,12,15-Octadecatrienoic acid, methyl ester, (Z,Z,Z)- structure (7.634%, RT 22.296) present in S. alata leaves |
 |
||||||||||||||||||||||||||||||||||||||||||||||||||
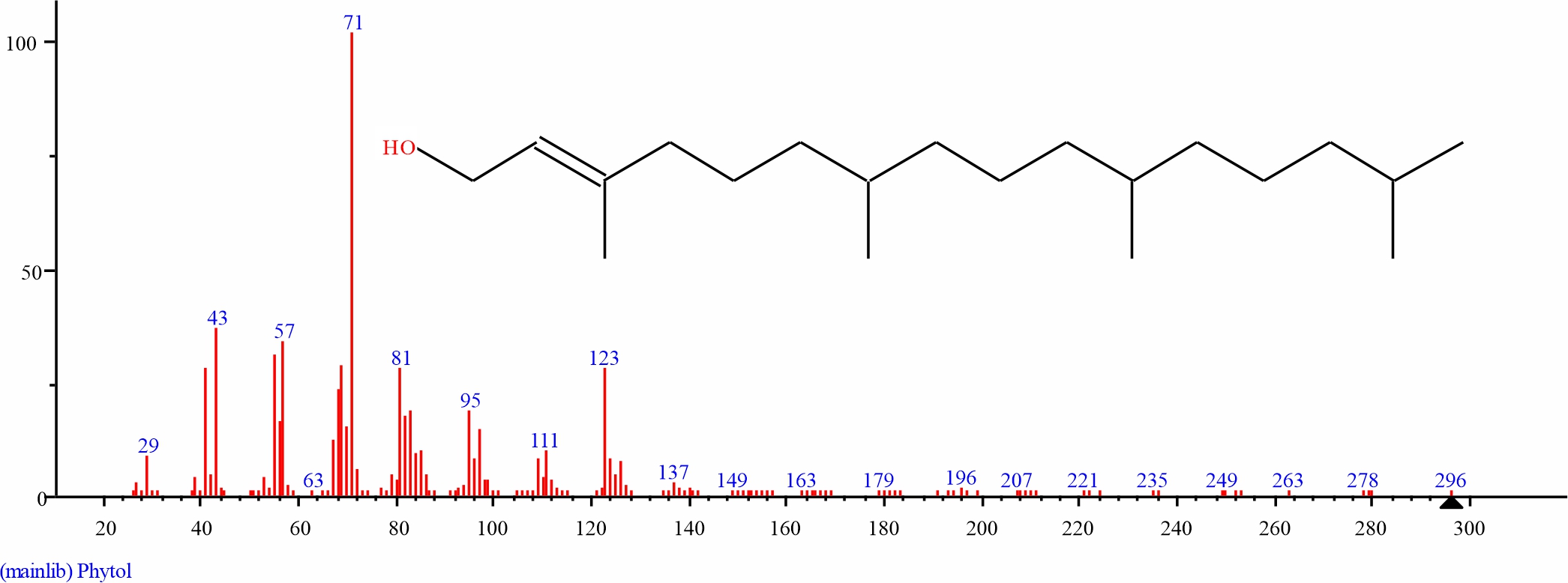
Fig. 7: mGC-MS spectra of the structure of Phytol (1.877%, RT 22.479) from S. alata leaves |
 |
||||||||||||||||||||||||||||||||||||||||||||||||||
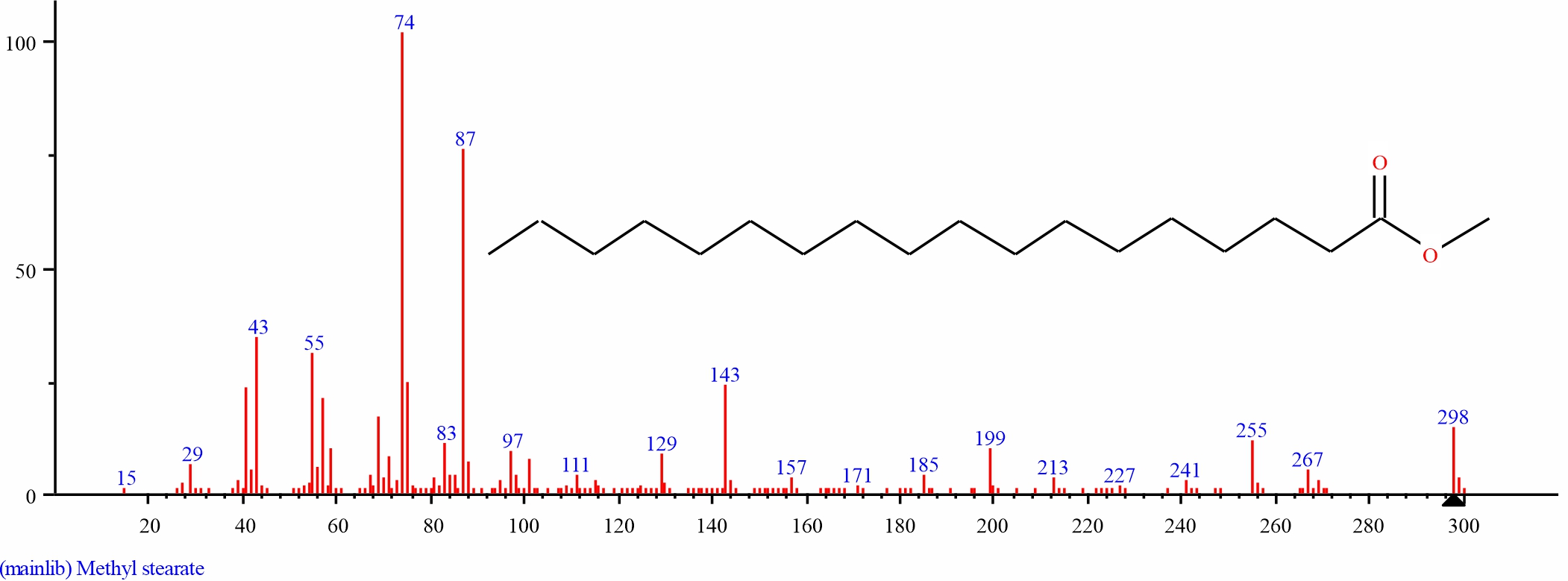
Fig. 8: GC-MS spectra of Methyl stearate structure (1.478%, RT 22.610) from S. alata leaves |
 |
||||||||||||||||||||||||||||||||||||||||||||||||||
Fig. 9: GC-MS spectra of 9,12-Octadecadienoic acid (Z,Z)- structure (5.283%, RT 23.211) from S. alata leaves |
 |
||||||||||||||||||||||||||||||||||||||||||||||||||
Fig. 10: GC-MS spectra of alpha Tocospiro A structure (3.021%, RT 31.222) from S. alata leaves |
 |
||||||||||||||||||||||||||||||||||||||||||||||||||
Fig. 11: GC-MS spectraof the structure of Nonacosane (4.487%, RT 31.319) from S. alata leaves |
 |
||||||||||||||||||||||||||||||||||||||||||||||||||
Fig. 12: GC-MS spectra of alpha Tocospiro B structure (4.317%, RT 31.405) from S. alata leaves |
 |
||||||||||||||||||||||||||||||||||||||||||||||||||
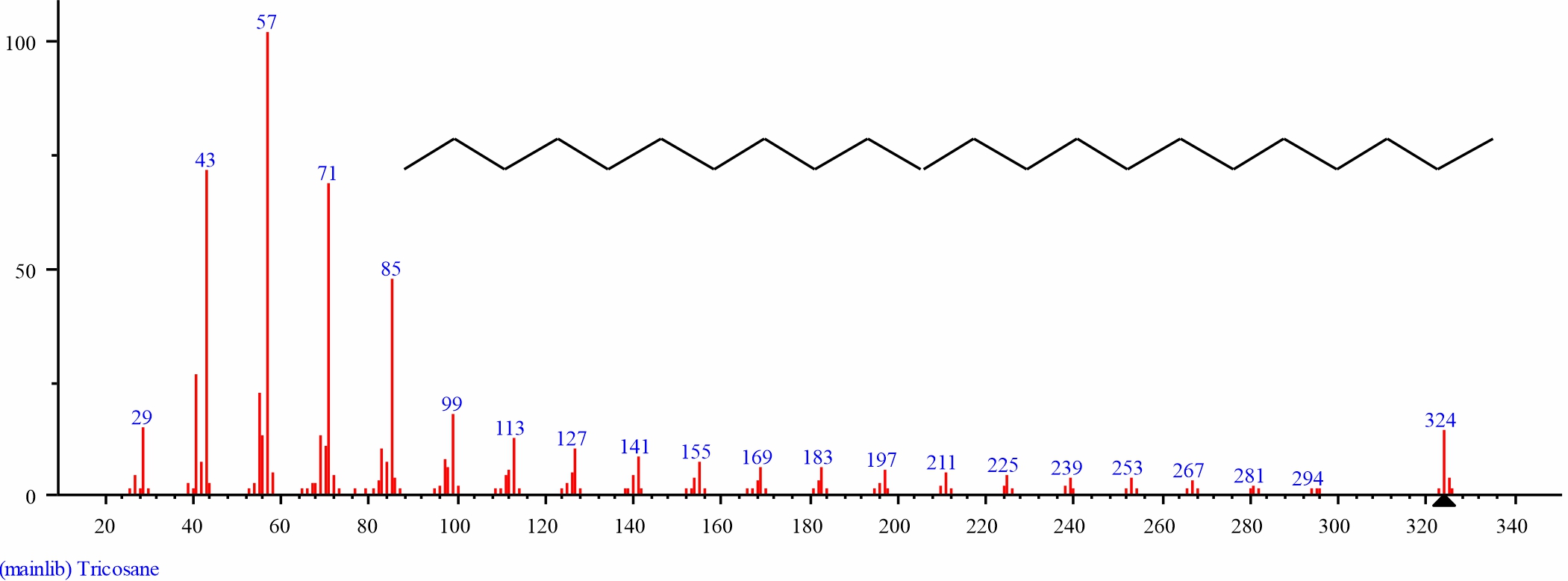
Fig. 13: GC-MS spectra of Tricosane structure (2.704%, RT 32.784) from S. alata leaves |
 |
||||||||||||||||||||||||||||||||||||||||||||||||||
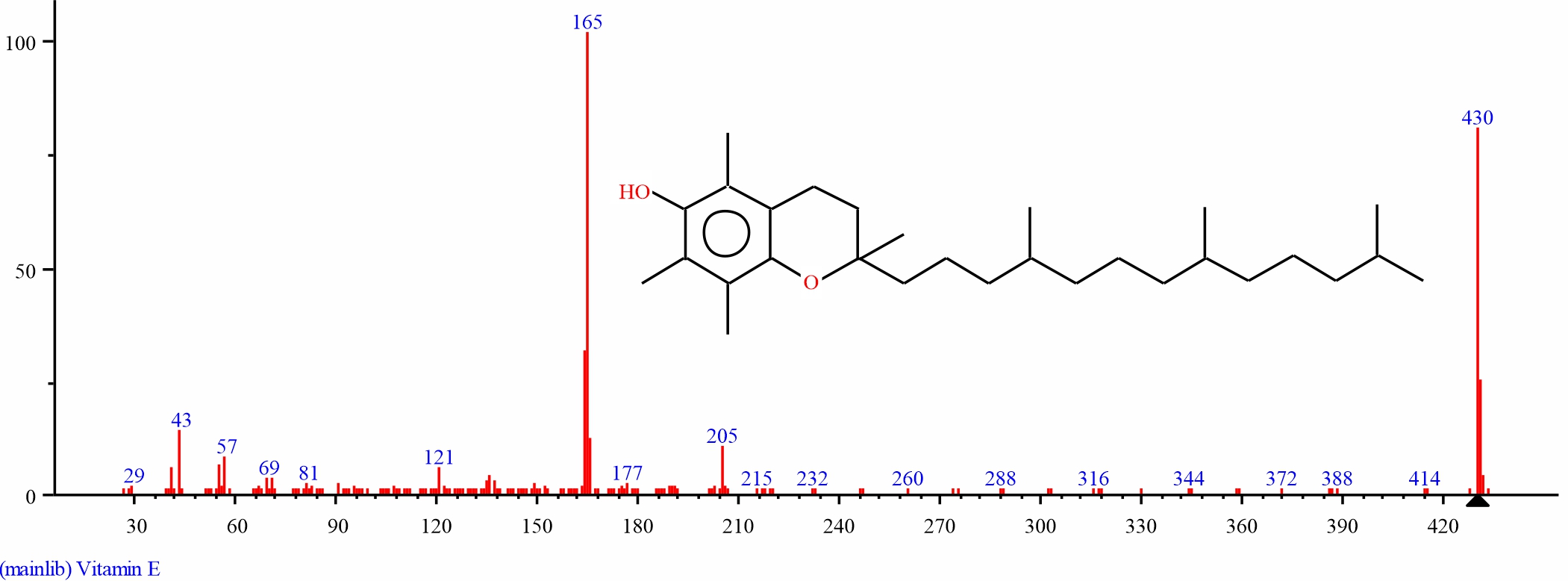
Fig. 14: GC-MS spectra of the structure of Vitamin E (38.338%, RT 33.419) from S. alata leaves |
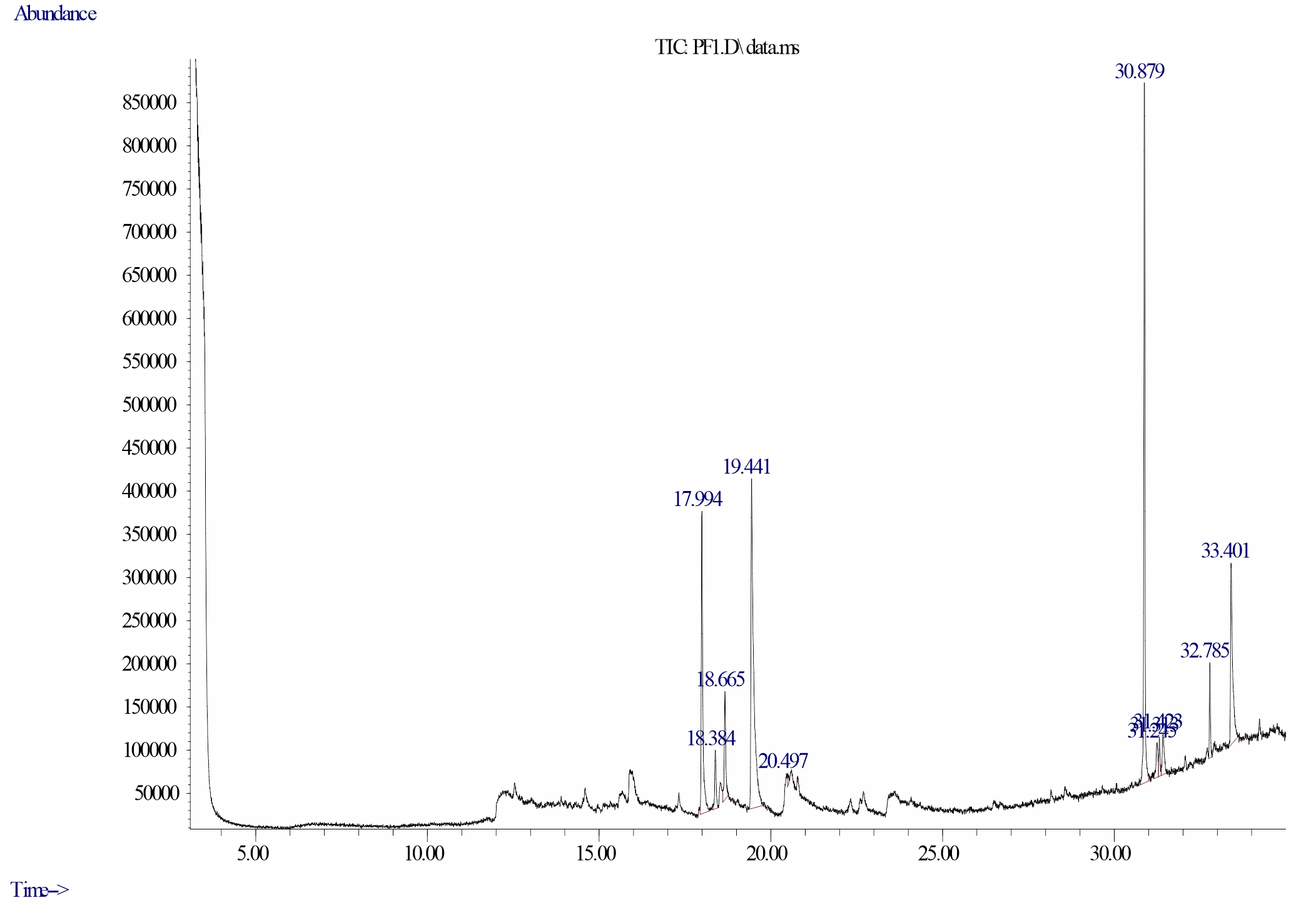
Gas chromatography-mass spectrometry analysis of dichloromethane extracts of leaves of D. tripetalla: The following thirteen bioactive compounds and their final peak values (Fig. 2) were observed in the leaf extract of D. tripetalla. They are Neophytadiene (14.793), 3, 7, 11, 15-Tetramethyl-2-hexadecen-1-ol (2.731), 3, 7, 11, 15-Tetramethyl-2-hexadecen- 1-ol (5.052), 5, 9, 13-Pentadecatrien-2-one, 6, 10, 14-trimethyl-, (E,E)- (27.778), n-Hexadecanoic acid (0.389), Supraene (27.917), α-Tocospiro B (2.021), Eicosyl isopropyl ether (1.545), α-Tocospiro A (2.630), 2-Methylpentacosane (3.044). and Vitamin E (12.099) (Table 2). The most abundant bioactive compounds identified in the chromatogram of dichloromethane leaf extracts of D.tripetalla are Supraene and 5, 9, 13-Pentadecatrien-2-one, 6, 10, at retention times of 30.879 and 19.441 minutes respectively while Eicosyl isopropyl ether and n-Hexadecanoic acid were the least observed at retention times of 31.3135 and 20.497 minutes respectively. The Retention Time, (min) Peak Area, Molecular formula and Molecular weight are shown in GC–MS spectra of the compounds are presented in Fig. 15-21.
 |
||||||||||||||||||||||||||||||||||||||||||||||||||
Fig. 15: Chromatogram of bioactive compounds in dichloromethane extract of D. tripetalla showing Supraene having a retention time of 30.879 minutes and final peak value of 27.917% |
|
|||||||
| S/N | Compound | Retention Time (min) | Peak Area (%) | Molecular formula | Molecular weight | ||
| 1 | Neophytadiene | 17.994 | 14.793 | C20H38 | 278.5157 | ||
| 2 | 3,7,11,15-Tetramethyl-2-hexadecen- 1-ol | 18.384 | 2.731 | C20H40O | 296.5310 | ||
| 3 | 3,7,11,15-Tetramethyl-2-hexadecen- 1-ol | 18.665 | 5.052 | C20H40O | 296.5310 | ||
| 4 | 5,9,13-Pentadecatrien-2-one, 6,10, 14-trimethyl-, (E,E)- | 19.441 | 27.778 | C18H30O | 262.4302 | ||
| 5 | n-Hexadecanoic acid | 20.497 | 0.389 | C16H32O2 | 256.4241 | ||
| 6 | Supraene | 30.879 | 27.917 | C30H50 | 410.7180 | ||
| 7 | alpha.-Tocospiro B | 31.245 | 2.021 | C29H50O4 | 462.715 | ||
| 8 | Eicosyl isopropyl ether | 31.313 | 1.545 | C23H48O | 340.63 | ||
| 9 | alpha.-Tocospiro A | 31.423 | 2.630 | C29H50O4 | 462.715 | ||
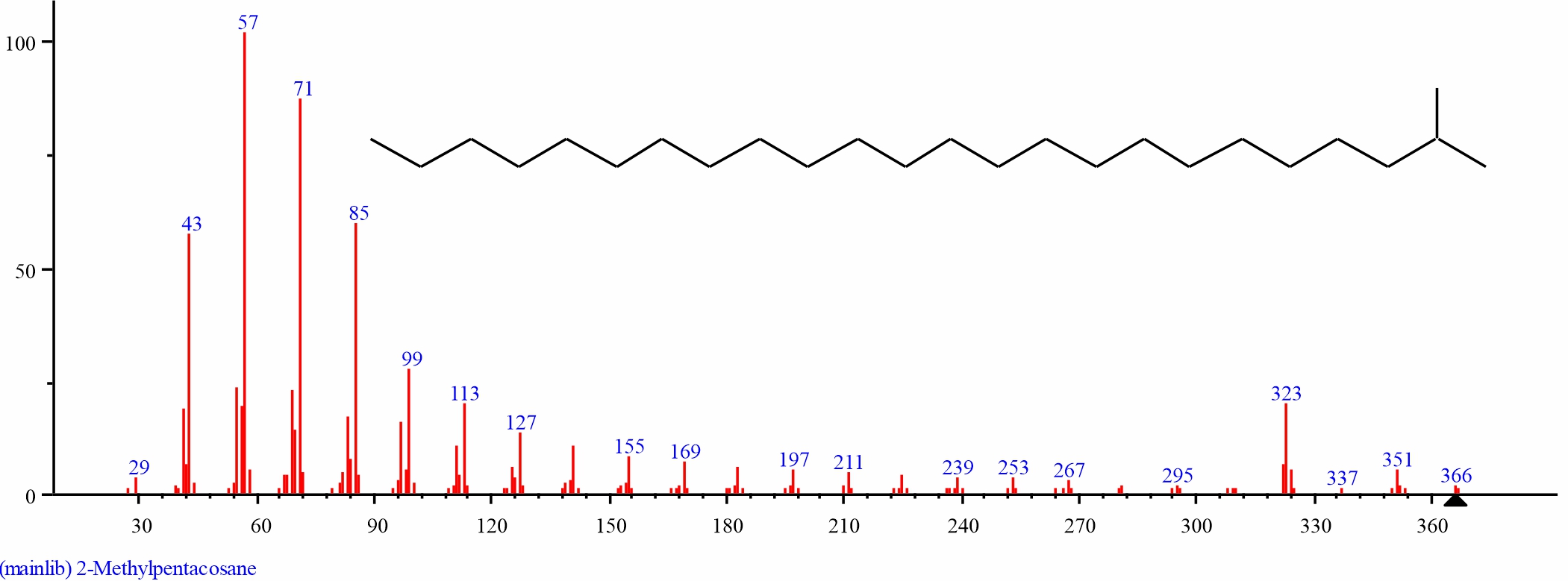
| 10 | 2-Methylpentacosane | 32.785 | 3.044 | C26H54 | 366.718 | ||
| 11 | Vitamin E | 33.401 | 12.099 | C29H50O2 | 430.717 | ||
 |
||||||||||||||||||||||||||||||||||||||||||||||||||
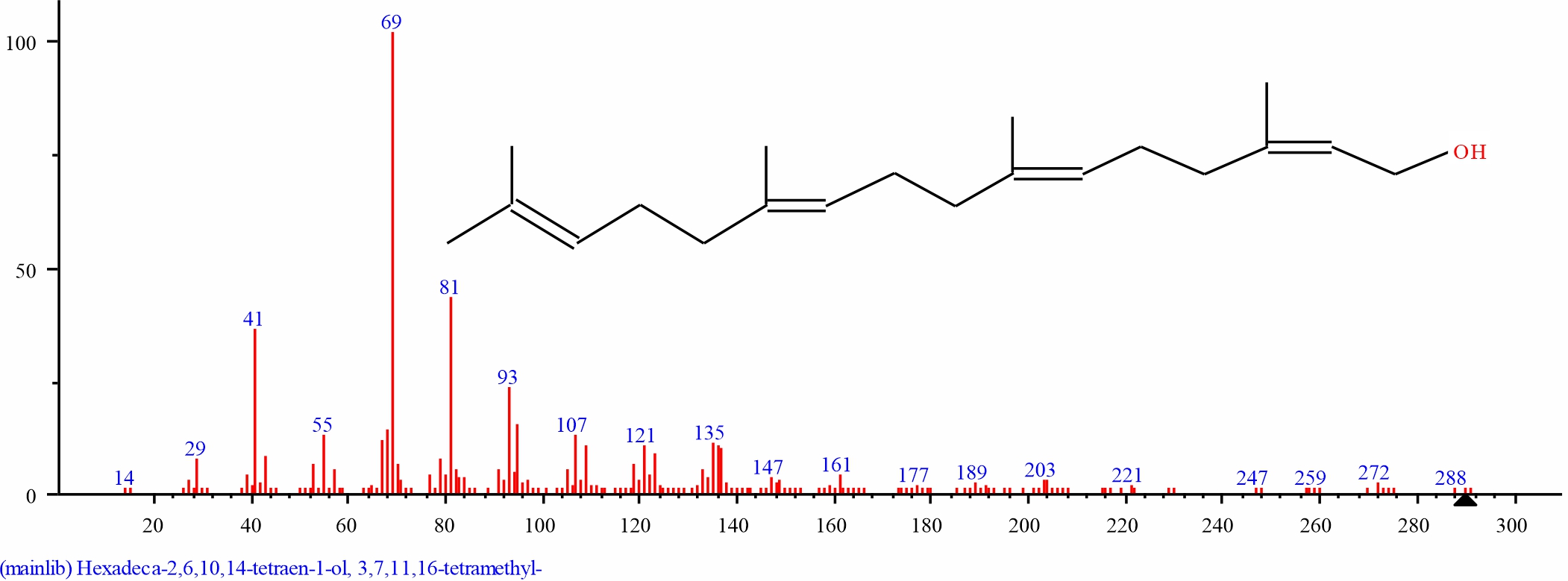
Fig. 16: GC-MS spectra of 3,7,11,15-Tetramethyl-2-hexadecen- 1-ol structure (2.732%, RT 18.384) from the leaves of D. tripetalla |
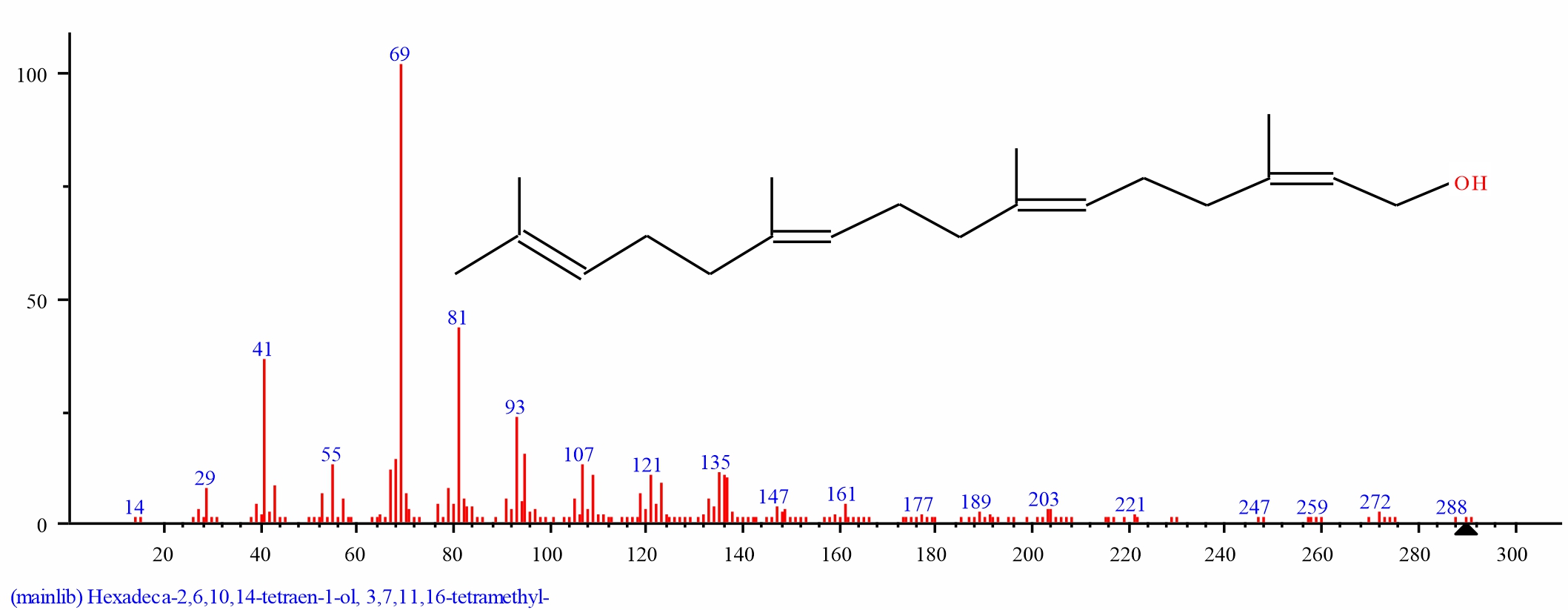 |
||||||||||||||||||||||||||||||||||||||||||||||||||
Fig. 17: GC-MS spectra of 3,7,11,15-Tetramethyl-2-hexadecen- 1-ol structure (5.052%, RT 18.665) from the leaves of D. tripetalla |
 |
||||||||||||||||||||||||||||||||||||||||||||||||||
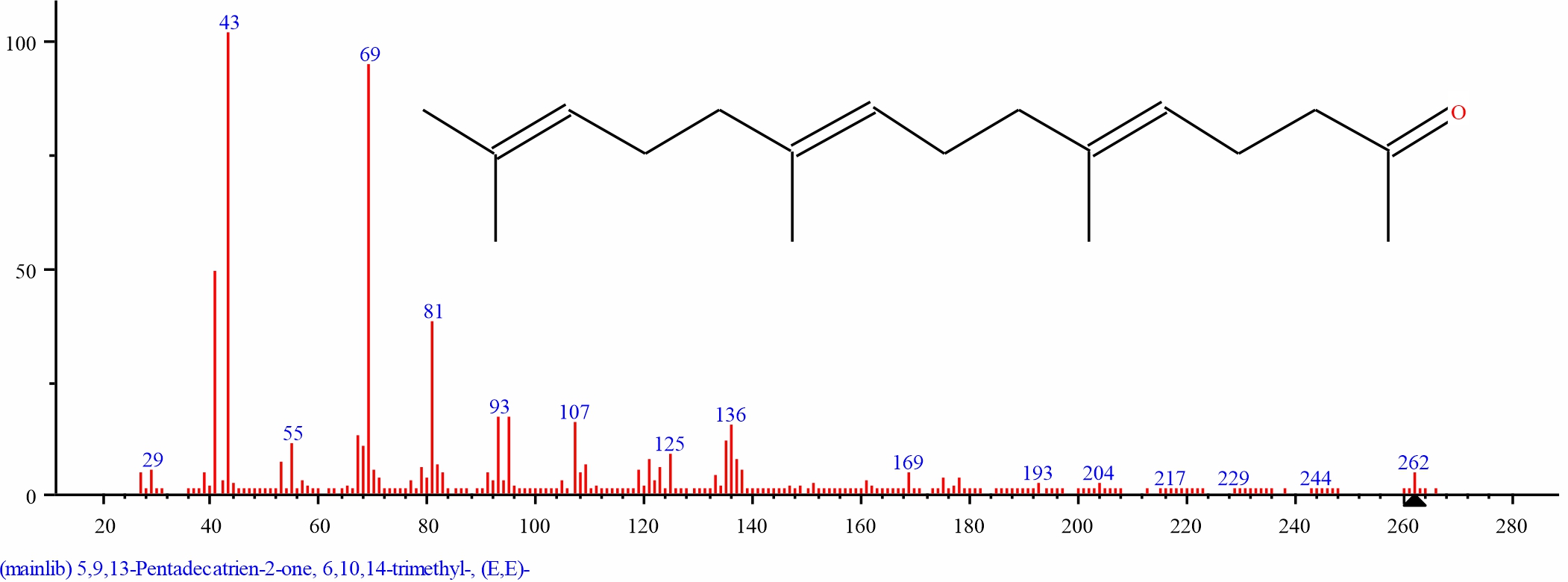
Fig. 18: GC-MS spectra of 5,9,13-Pentadecatrien-2-one, 6,10, 14-trimethyl-, (E,E)- structure (27.778%, RT 19.441) from the leaves of D. tripetalla |
 |
||||||||||||||||||||||||||||||||||||||||||||
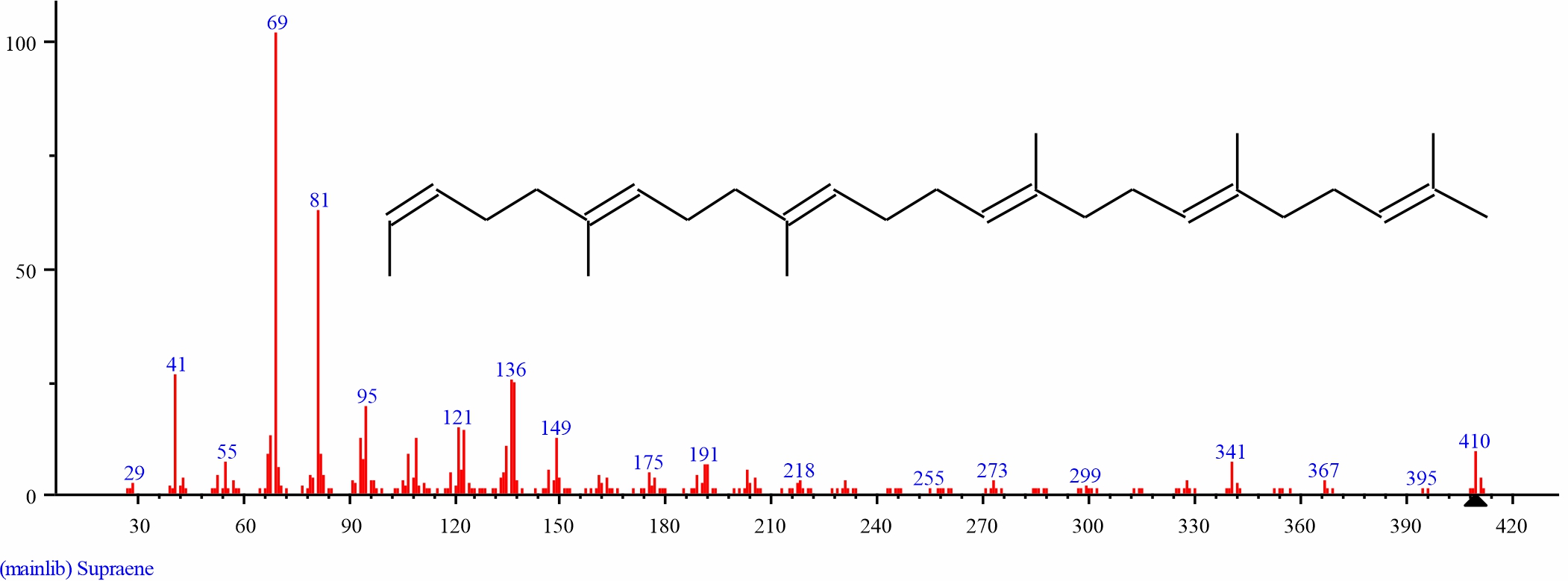
Fig. 19: GC-MS spectra of Supraene structure (27.917%, RT 30.879) from the leaves of D. tripetalla |
 |
||||||||||||||||||||||||||||||||||||||||||||
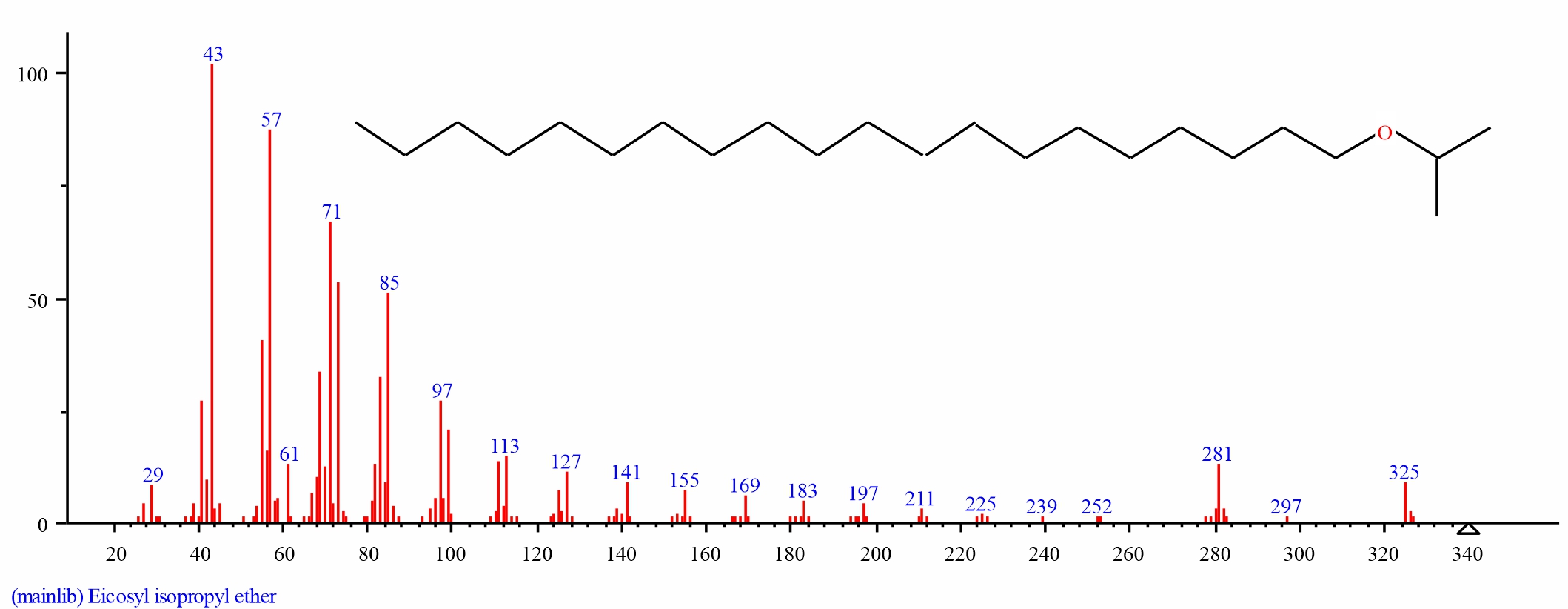
| Fig. 20: GC-MS spectra of Eicosyl isopropyl ether structure (1.545%, RT 31.313) from the leaves of D. tripetalla |
 |
||||||||||||||||||||||||||||||||||||||||||||
Fig. 21: GC-MS spectra of 2-Methylpentacosane structure (3.044%, RT 32.785) from the leaves of D. tripetalla |
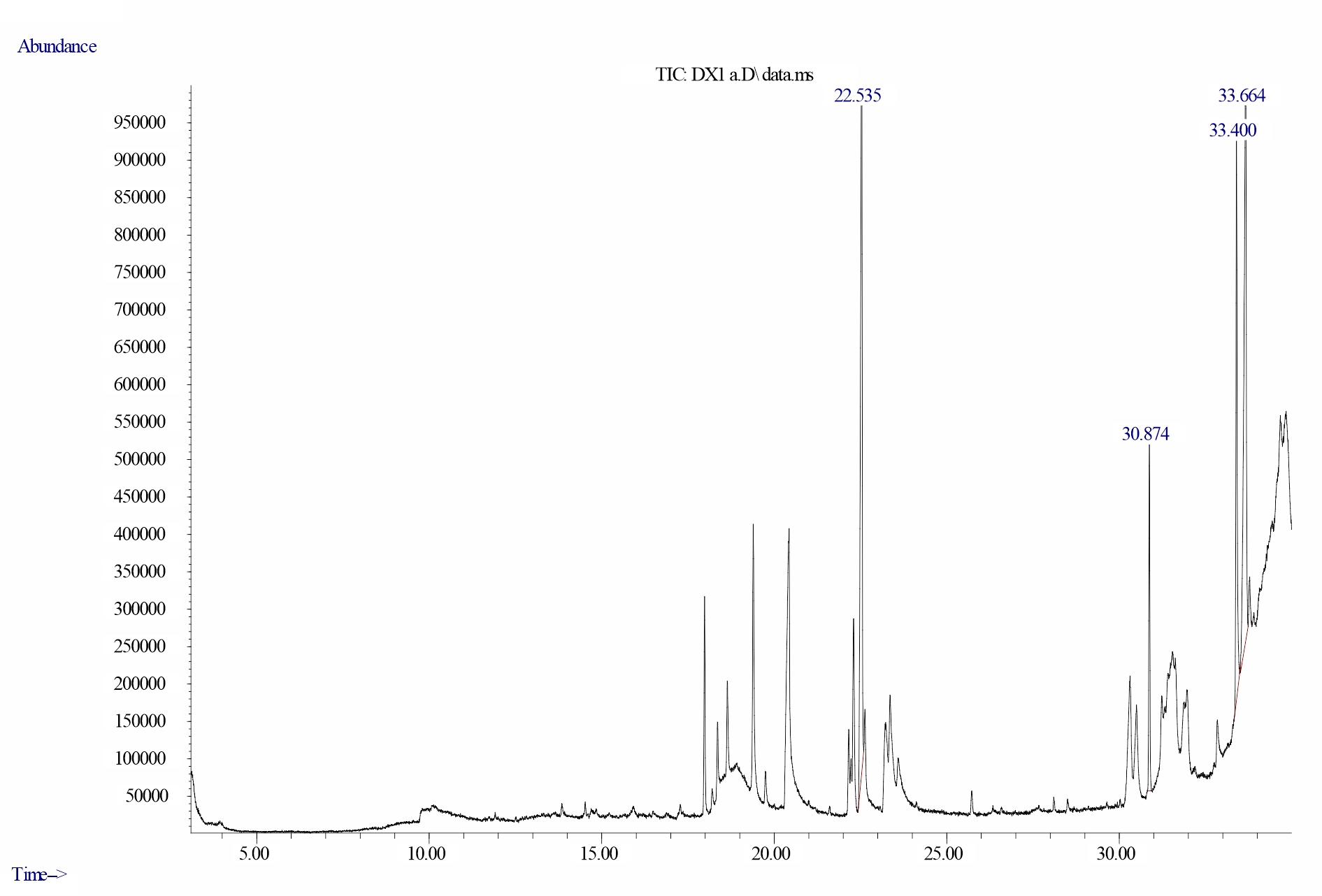
Gas chromatography-mass spectrometry analysis of dichloromethane extracts of Delonixregia leaves: Analysis of data from the GC–MS spectrum of dichloromethane leaf extracts of D. regia revealed the presence of four bioactive compounds of biological importance as shown in Figures 22-24 and Table 3. These are Dodecanoic acid, 1, 2, 3-propanetriyl ester, Phytol, Vitamin E, and Squalene. Highest peak of was observed for Dodecanoic acid, 1, 2, 3-propanetriyl ester (37.228%), Phytol (34.681%) and vitamin E (18.786%) at retention times of 33.664, 22.535 and 33.400 minutes respectively while Squalene had the least peak (9.305%) at a retention time of 30.874 minutes.
 |
|||||||
Fig. 22: Chromatogram of bioactive compounds in dichloromethane extract of D. regia showing Dodecanoic acid, 1,2,3-propanetriyl ester having a retention time of 33.664 minutes and final peak value of 37.228% |
|
|||||||
| S/N | Compound | Retention Time (min) | Peak Area (%) | Molecular formula | Molecular weight | ||
| 1 | Phytol | 22.535 | 34.681 | C20H40O | 296.539 | ||
| 2 | Squalene | 30.874 | 9.305 | C30H50 | 410.718 | ||
| 3 | Vitamin E | 33.400 | 18.786 | C29H50O2 | 430.7100 | ||
| 4 | Dodecanoic acid, 1,2,3-propanetriyl ester | 33.664 | 37.228 | C39H74O6 | 639.0013 | ||
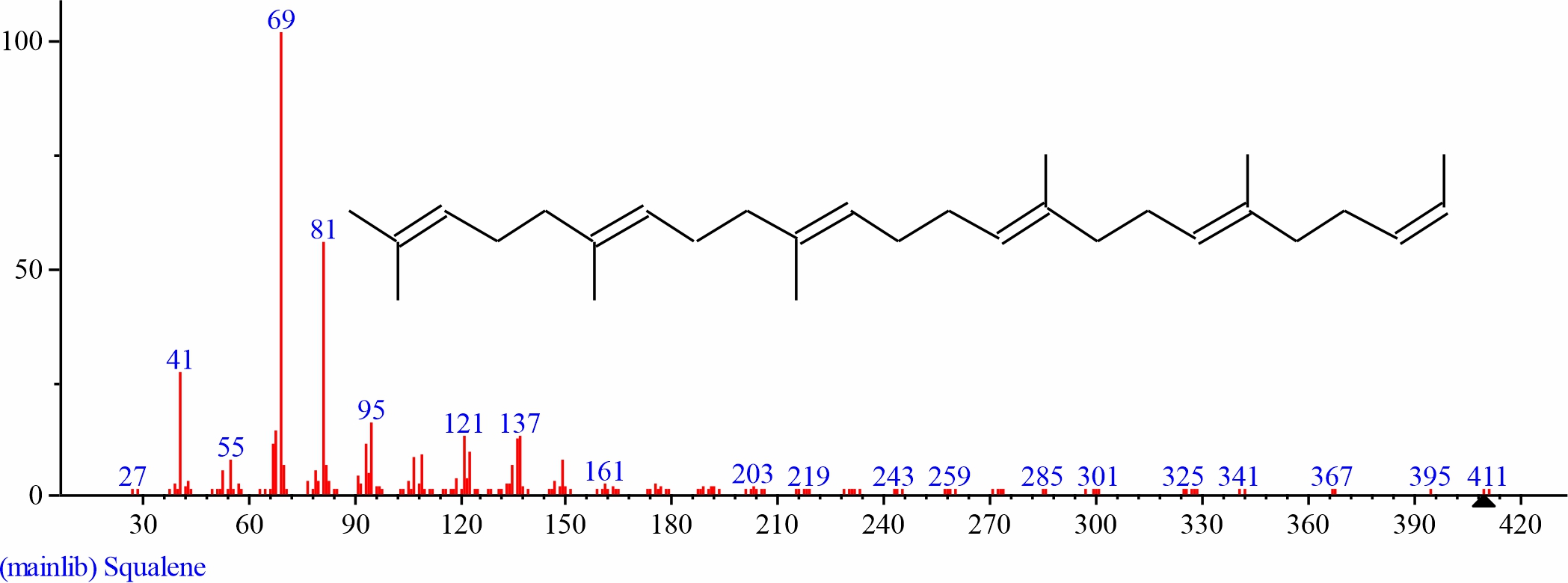 |
Fig. 23: GC-MS spectra of Squalene structure (9.305%, RT 30.874) from D. regia leaves |
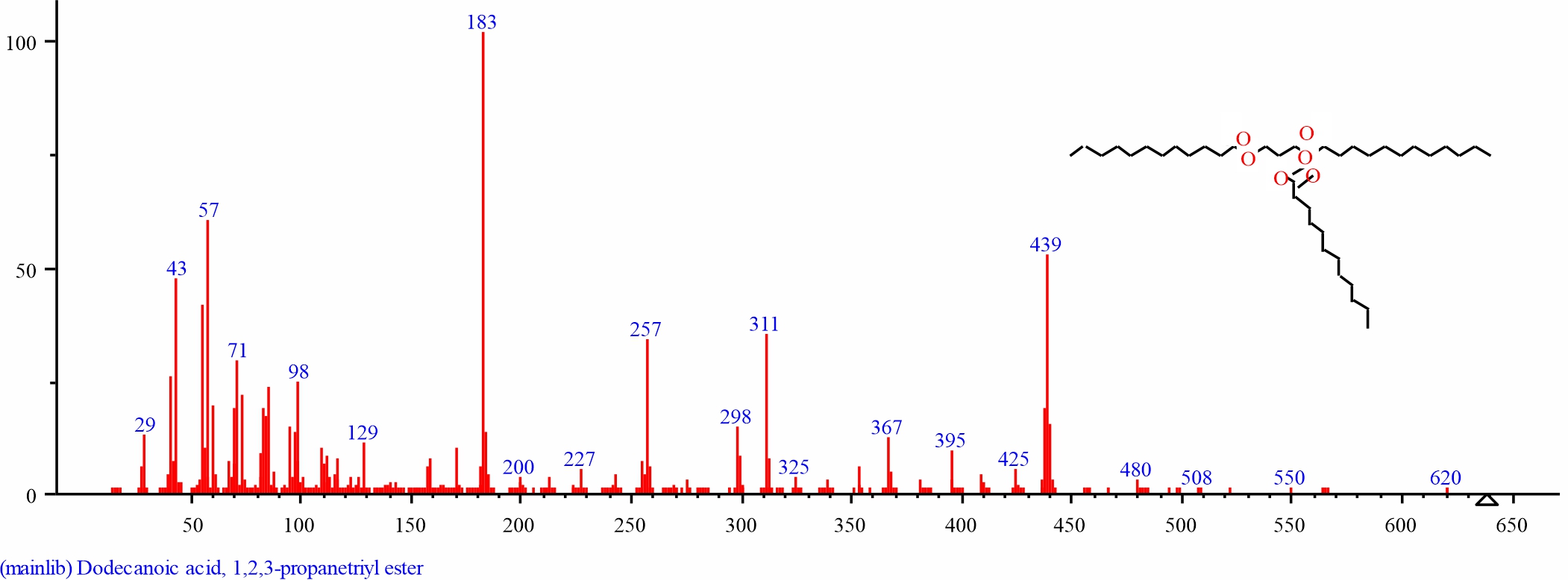 |
Fig. 24: GC-MS spectra of Dodecanoic acid, 1,2,3-propanetriyl ester structure (37.228%, RT 33.664)from D. regia leaves |
The nutritional quality of plants may be evaluated for their phytochemical and mineral composition. However, biochemical analysis of the plant using a GC-MS will further revealthe presence of bioactive compounds present in these plants, elucidate their structures as well as indicate their molecular weights. GC-MS analysis of dichloromethane extracts of S. alata, D. tripetalla, and D. regia leaves revealed the presence of bioactive compounds that have been shown to possess pharmacologic activities which may be responsible for the traditional use of these plants for the treatment of various illnesses. For example, hexadecanoic acid methyl ester was detected in S. alata and D. tripetalla. It is a fatty acid with antioxidant, anti-inflammatory, antihyperlipidemic, antimicrobial, pesticidal, anti-androgenic and nematicidal effects. It is used as a flavoring agent and a lubricant33-34. n-Hexadecanoic acid is a fatty acid and was confirmed to exhibit anti-inflammatory35 antioxidant, nematicide, pesticidal, anti-androgenic, hemolytic, 5-alpha reductase inhibiting36, mosquito larvicidal effects37. Phytol is acyclic diterpene alcohol commonly referred to as isoprenoids, which is generally produced from the degradation of chlorophyll. Phytol was detected in the leaf extracts of S. alataand D. regia. It was proven to exhibit strong antioxidant and antinociceptive activities antimalarial, antimicrobial, antifungal, anti-inflammatory and also has diuretic property38-40. It is also reported to be effective against S. typhi, resistant gonorrhea, joint dislocation, headache, hernia, and is used as a stimulant41. Phytol is also a precursor of synthetic vitamin E and vitamin K1, it was found to be cytotoxic against breast cancer cell lines (MCF7)42-43. Methyl stearate is used as a solvent or cosolvent and lipid vesicle in agriculture. A fatty acid derivative, 9,12-octadecadienoic acid, methyl ester (Z, Z) has been reported to possess strong anticancer properties44. This may probably account for the cytotoxicity observed in the ethyl acetate extract of Senna alata (L) Roxb against some human carcinomas - MCF 7 (human breast), C4-2WT (prostate), HT 29 and HTC 116 (colorectal) cell lines45. Ponnamma and Manjunath46 had earlier reported anti-inflammatory, hypocholesterolemic and antiarthritic activity of 9,12-octadecadienoic acid, methyl ester (Z, Z). An unsaturated fatty acid, 9, 12, 15-Octadecatrienole acid (Z, Z, Z) is a lipid metabolism regulator reported to have anti-inflammatory, hypocholesterolemic, cancer preventive, hepatoprotective, nematicide, insectifuge, antihistamininic, antieczemic, 5-alpha reductase inhibitor, antiandrogenic, anti-arthritic, and anti-coronary properties47. Squalene was detected in D. regia. It is asaturated fatty acid, a hexaisoprenoid (triterpenoid) hydrocarbon; intermediate in the biosynthesis of cholesterol and other sterols and triterpenes. They are found in shark oil and some plants. It is a volatile compound reported to have antibacterial, antioxidant, antitumor and cancer preventive properties immune stimulant potentials. It was also found to be a potent lipoxgenase inhibitor and a pesticide48-49. Similarly, Dodecanoic acid, 1, 2, 3-propanetriyl ester found present in the leaf extract of D. regia has been reported by Taskova et al.50 to possess antibacterial, antioxidant, antiviral, candidiasis and hypercholesterolemic properties. The result of the extractions also showed the presence of alpha-Tocospiro A and B, as well as vitamin E. Vitamin E, particularly alpha-tocopherol acts as an antioxidant by reacting with the free radicals and converts them to harmless substances. In addition to its antioxidant activity, alpha-tocopherol is also suggested to play a role in immune enhancement, inhibition of platelet aggregation and anti-inflammatory functions51-52. Research indicates the presence of Neophytadiene in the leaf extracts of S. alata and D. tripetalla. Neophytadiene, a sesquiterpenoid is a good analgesic, antipyretic, anti-inflammatory, antimicrobial, and antioxidant compound. It also has carminative, gastrin inhibitor, antiulcerative, histamine release inhibitor, antiprotozoal (leishmania) and antiparasiticproperties53-54. Nonacosane was detected in the leaf extract of S. alata. It is a hydrocarbon that plays a role in the chemical communication of several insects, for example, it is known to be a pheromone of the female Anopheles stephensi mosquito55. Supraenes were detected in D. tripetalla extract. They are very long-chain polyunsaturated fatty acids (VLC-PUFAs) so-called to indicate the presence of more than six double bonds per molecule and are present in all four glycerophospholipids. They are important components of retinal phospholipids, they form less than 15% of all the phosphoglycerides and are probably incorporated into retinal membranes56. ß-Citronellol methyl ether was detected in the leaf extract of S. alata. It is a monoterpene alcohol that has been shown to confer fragrance, aroma, and taste to flowering plants57-58.
CONCLUSION
The analysis of this work revealed the presence of major bioactive compounds in the dichloromethane leaf extracts of S. alata, D. tripetalla and D. regia which have also been reported to play crucial roles in disease and general metabolisms of humans and therefore support the traditional medicinal application of these plants. Each of these bioactive compounds can further be extracted, and its pharmacological activity clinically evaluated for possible drug development.
SIGNIFICANCE STATEMENT
This study discovered that the dichloromethane leaf extracts of S. alata, D. tripetalla and D. regia contain bioactive compounds that can be beneficial as therapeutic agents for management of various illnesses. This study will help the researchers to uncover the critical areas of traditional medicinal application that many researchers were not able to explore. Thus a new theory on the use of medicinal plant extracts in the management of diseases may be arrived at.
REFERENCES
- Mgbeahuruike, E.E., T. Yrjönen, H. Vuorela and Y. Holm, 2017. Bioactive compounds from medicinal plants: focus on piper species. South Afr. J. Bot., 112: 54-69.
- Sarangapani, S. and M. Rajappan, 2012. Pharmacognostical and pharmaceutical characterization of delonixregia - a novel matrix-forming natural polymer. Int. J. Pharm., 2: 564-573.
- Egharevba, , H.O. , Ibrahim, J.A. and Nduaguba et al., 2015. Phytochemical, proximate pharmacognostic analyses and thin layer chromatography of chrysophyllumalbidum seed. Ew. J. Herb. Chem. Pharmacol. Res., 1: 6-12.
- Atanasov, A.G., B. Waltenberger, E.M. Pferschy-Wenzig, T. Linder and C. Wawrosch et al., 2015. Discovery and resupply of pharmacologically active plant-derived natural products: A review. Biotechnol. Adv., 33: 1582-1614.
- Guo, F., L. Feng, C. Huang, H. Ding, X. Zhang, Z. Wang and Y. Li, 2013. Prenylflavone derivatives from Broussonetia papyrifera, inhibit the growth of breast cancer cells in vitro and in vivo. Phytochem. Lett., 6: 331-336.
- Momin, M.A.M., S.F. Bellah, S.M.R. Rahman, A.A. Rahman, G.M.M. Murshid and T.B. Emran, 2014. Phytopharmacological evaluation of ethanol extract of Sida cordifolia L. roots. Asian Pac. J. Trop. Biomed., 4: 18-24.
- Farid, M.M., S.R. Hussein, L.F. Ibrahim, M.A.E. Desouky, A.M. Elsayed, A.A.E. Oqlah and M.M. Saker, 2015. Cytotoxic activity and phytochemical analysis of arum palaestinum boiss. Asian Pac. J. Trop. Biomed., 5: 944-947.
- Roskov, Y., L. Abucay, T. Orrell, D. Nicolson and N. Bailly et al., 2018. Species 2000 and ITIS Catalogue of Life, 2018 Annual Checklist. Digital resource at www.catalogueoflife.org/annual-checklist/2018. Species 2000: Naturalis, Leiden, the Netherlands.
- Bikovens, O., L. Roze, A. Pranovich, M. Reunanen and G. Telysheva, 2013. Chemical composition of lipophilic extractives from grey alder (alnusincana). BioResources, 8: 350-357.
- Veigas, J.M., P. Divya and B. Neelwarne, 2012. Identification of previously unreported pigments among carotenoids and anthocyanins in floral petals of Delonix regia (hook.) raf. Food Res. Int., 47: 116-123.
- Adewuyi, A., R.A. Oderinde, B.V.S.K. Rao, R.B.N. Prasad and B. Anjaneyulu, 2010. Chemical component and fatty acid distribution of Delonix regia and Peltophorum pterocarpum seed oils. Food Sci. Technol. Res., 16: 565-570.
- Salem, M.Z.M., 2013. Evaluation of the antibacterial and antioxidant activities of stem bark extracts of Delonix regia and Erythrina humeana grown in Egypt. J. For. Prod. Ind., 2: 48-52.
- Rosmalena, F., A. Amalia, I. Syafitri and V.D. Prasasty, 2017. Antimalarial effect of flamboyant (Delonix regia) bark and papaya (Carica papaya L.) leaf ethanolic extracts against Plasmodium berghei in mice. Biomed. Pharmacol. J., 10: 1081-1089.
- Ogboye, P.O., B.M. Onyegeme-Okerenta and C. Monago-Ighorodje, 2018. Proximate composition, antioxidant and hypoglycaemic potential of aqueous extracts of seeds of Delonix regia on high fat diet and streptozotocin-induced diabetes in female wistar rats. Clin. Exp. Med. Sci., 6: 33-46.
- Jahan, I., M. Rahman, M. Rahman, M. Kaisar, M. Islam, A. Wahab and M. Rashid, 2010. Chemical and biological investigations of Delonix regia (bojer ex hook.) raf. Acta Pharm., 60: 207-215.
- Shabir, G., F. Anwar, B. Sultana, Z.M. Khalid, M. Afzal, Q.M. Khan and M. Ashrafuzzaman, 2011. Antioxidant and antimicrobial attributes and phenolics of different solvent extracts from leaves, flowers and bark of gold mohar [Delonix regia (Bojer ex Hook.) Raf.]. Molecules, 16: 7302-7319.
- Parekh, J., D. Jadeja and S. Chanda, 2005. Efficacy of aqueous and methanol extracts of some medicinal plants for potential antibacterial activity. Turk. J. Biol., 29: 203-210.
- Adedayo, O., W.A. Anderson, M. Young, V. Snieckus, P.A. Patil and D.O. Kolawole, 2001. Phytochemistry and antibacterial activity of Senna alata flower. J. Pharm. Biol., 39: 408-412.
- Hennebelle, T., B. Weniger, H. Joseph, S. Sahpaz and F. Bailleul, 2009. Senna alata. Fitoterapia, 80: 385-393.
- Ajibesin, K.K., B.J. Ekpo, D.N. Bala, E.E. Essien and S.A. Adesanya, 2008. Ethnobotanical survey of akwa ibom state of Nigeria. J. Ethnopharmacol., 115: 387-408.
- Onyegeme-Okerenta, B.M., T. Nwosu and M.O. Wegwu, 2017. Investigation of the effect of aqueous leaf extract of Senna alata (L) roxb on palm oil-induced hyperlipidaemic plasma lipid in wister rats. Clin. Exp. Med. Sci., 5: 19-34.
- Ejechi, B.O. and D.E. Akpomedaye, 2005. Activity of essential oil and phenolic acid extracts of pepperfruit (dennetiatripetala G. barker; anonaceae) against some food-borne microorganisms. Afr. J. Biotechnol., 4: 258-261.
- Anyaele, O.O. and A.A.S. Amusan, 2003. Toxicity of hexanolic extract of Dennettia tripetala on larvae of Aedes aegypti. Afr. J. Biomed. Res., 6: 49-53.
- Anaga, A.O. and I.U. Asuzu, 2009. Antihyperglycaemic properties of the ethyl acetate extract of Dennetia tripetala in diabetic rats. J. Complem. Integ. Med.
- Oyemitan, I.A., E.O. Iwalewa, M.A. Akanmu and T.A. Olugbade, 2008. Antinociceptive and antiinflammatory effects of essential oil of Dennettia tripetala G.Baker (Annonaceae) in rodents. Afr. J. Trad. Compl. Alt. Med., 5: 355-362.
- Okolie, N., A. Falodun and O. Davids, 2014. Evaluation of the antioxidant activity of root extract of pepper fruit (Dennetia tripetala), and it`s potential for the inhibition of lipid peroxidation. Afr. J. Trad., Compl. Alt. Med., 11: 221-227.
- Idoko, J.E. and R.A. Adebayo, 2011. Effect of dennettia tripetala powder and reduced rates of pirimiphos-methyl singly and combined in management of Callosobruchus maculatus (f.) [coleoptera: bruchidae]. Int. J. Biol., 3: 174-178.
- Ihemeje, A., M.C. Ojinnaka, K.C. Obi and C.C. Ekwe, 2013. Biochemical evaluation of pepperfruit (Dennettia tripetala) and its use as substitute for ginger in zobo drink production. Acad. Res Int.,, 4: 513-521.
- Onyegeme-Okerenta, B. and F. Anacletus, 2017. Hypoglycaemic and hypolipidaemic potentials of senna alata and its effect on the pancreas of alloxan-diabetic induced albino rats. J. Appl. Life Sci. Int., 11: 1-10.
- Omeodu, S.I., B.M. Onyegeme-Okerenta and A.P. Amakiri, 2017. Effect of Delonix regia seed extract on some liver enzyme markers and electrolytes of alloxan-induced diabetic rats. J. Pharm. Sci. Innovation, 6: 37-43.
- Onyegeme-Okerenta, B.M., B.A. Amadi and V.O. Ezeonyilimba, 2018. The ameliorating potential of Annona muricata on sodium fluoride-induced toxicity on liver and kidney of male wistar rats. J. Compl. Alt. Med. Res., 6: 1-17.
- Ukwubile, C.A., A. Ahmed, U.A. Katsayal, J. Ya'u and S. Mejida, 2019. GC-MS analysis of bioactive compounds from Melastomastrum capitatum (Vahl) Fern. leaf methanol extract: An anticancer plant. Scient. Afr.
- Ibibia, E.T., K.M. Olabisi and O.S. Oluwagbemiga, 2016. Gas chromatography-mass spectrometric analysis of methanolic leaf extracts of lanneakerstingii and naucleadiderrichii, two medicinal plants used for the treatment of gastrointestinal tract infections. Asian J Pharm Clin Res., 9: 179-182.
- Pramitha, V.S. and N.S. Kumari, 2016. Anti-inflammatory, antioxidant, phytochemical and GC-MS analysis of marine brown macroalga, sargassumwighti, Int. J. Pharm. Chem. Biol. Sci., 6: 7-15.
- Sermakkani, M. and V. Thangapandian, 2012. GC-MS analysis of cassia italica leaf methanol extract. Asian J. Pharm. Clin. Res., 5: 90-94.
- Jayamathi, T., N. Komalavalli and V. Pandiyarajan, 2012. GC-MS analysis of leaf ethanolic extracts of Wrightia tinctoria - a high medicinal value plant. Asian J. Plant Sci. Res., 2: 688-691.
- Thomas, E., T.P. Aneesh, D.G. Thomas and R. Ananda, 2013. GC-MS analysis of phytochemical compounds present in rhizomes of nerviliaaragoana gaud. Asian J. Pharm. Clin. Res., 6: 68-74.
- Santos, C., M. Salvadori, V. Mota, L. Costa and A. de Almeida et al., 2013. Antinociceptive and antioxidant activities of phytol in vivo and in vitro models. Neurosci. J.
- Pejin, B., A. Savic, M. Sokovic, J. Glamoclija and A. Ciric et al., 2014. Further in vitro evaluation of antiradical and antimicrobial activities of phytol. Nat. Prod. Res., 28: 372-376.
- De Moraes, J., R.N. de Oliveira, J.P. Costa, A.L. Junior and D.P. de Sousa et al., 2014. Phytol, a diterpene alcohol from chlorophyll, as a drug against neglected tropical disease Schistosomiasis mansoni. PLoS Negl. Trop. Dis.
- Dandekar, R., Fegade, B., Bhaskar, V.H., 2015. GC-MS analysis of phytoconstituents in alcohol extract of Epiphyllum oxypetalum leaves. J. Pharmacogn. Phytochem., 4: 149-154.
- Ogunlesi, M, W. Okiei, E. Ofor and A.E. Osibote, 2009. Analysis of the essential oil from the dried leaves of Euphorbia hirta Linn (Euphorbiaceae), a potential medication for asthma. Afr. J. Biotechnol., 8: 7042-7050.
- Satyal, P., N.S. Dosoky, A. Poudol and W.N. Setzer, 2012. Essential oil constituents and their biological activities from the leaves of Cassia fistula growing in Nepal. Open Access J. Med. Aromat Plants, 3: 1-4.
- Klotz, L.G.A., 1942. A simplified method of growing plants with roots in nutrient vapors. Phytopathology, 34: 507-508.
- Onyegeme-Okerenta, B., 2018. Ethyl acetate extract of Senna alata (l) roxb increases cytotoxicity in the human breast, prostate and colorectal cancer cells. J. Cancer Treat. Res., 6: 44-53.
- Ponnamma, S.U. and K. Manjunath, 2012. GC-MS analysis of phytocomponents in the methanolic extract of Justicia wynaadensis (NEES) T. Anders. Int. J. Pharma Bio Sci., 3: 570-576.
- Tyagi, T. and M. Agarwal, 2017. Phytochemical screening and GC-MS analysis of bioactive constituents in the ethanolic extract of Pistia stratiotes L. and Eichhornia crassipes (Mart.) solms. J. Pharmacogn. Phytochem., 6: 195-206.
- Agnel, R.A. and V.R. Mohan, 2014. GC-MS analysis of bioactive compounds present in the whole plant of Andrographis echioides (linn.) nees (acanthaeceae). Eur. J. Biomed. Pharm., 1: 443-452.
- Kesava, R.B. and R.G. Usha, 2016. GC-MS analysis of volatile components in petroleum ether extracts of Coldenia procumbens linn. Int. J. Pharm. Bio. Sci., 7: 241-245.
- Taskova, R., M. Mitova, B. Mikhova and H. Duddeck, 2003. Bioactive phenolics from Carthamus lanatus L. Z. Naturforsch. C, 58: 704-707.
- Jensen, S.K. and C. Lauridsen, 2007. ??tocopherol stereoisomers. Vitamin E, 76: 281-308.
- Chen, C.R., L.H. Chao, Y.W. Liao, C.I. Chang and M.H. Pan, 2007. Tocopherols and triterpenoids from Sida acuta. J. Chin. Chem. Soc., 54: 41-45.
- Venkata, B., L.A. Raman, M. Samuel and S. Pardha, 2012. Antibacterial, antioxidant activity and GC-MS analysis of eupatorium odoratum. Asian J. Pharm. Clin. Res., 5: 99-106.
- Mustapa, A.N., A. Martin, R.B. Mato and M.J. Cocero, 2015. Extraction of phytocompounds from the medicinal plant Clinacanthus nutans Lindau by microwave-assisted extraction and supercritical carbon dioxide extraction. Ind. Crops Prod., 74: 83-94.
- Brei, B., J.D. Edman, B. Gerade and J.M. Clark, 2004. Relative abundance of two cuticular hydrocarbons indicates whether a mosquito is old enough to transmit malaria parasites. J. Med. Entomol., 41: 807-809.
- Aveldaño, M.I., S.J.P. de Garcia and N.G. Bazán, 1983. Biosynthesis of molecular species of inositol, choline, serine, and ethanolamine glycerophospholipids in the bovine retina.. J. Lipid Res., 24: 628-638.
- Zhao, C.Y., J. Xue, X.D. Cai, J. Guo, B. Li and S. Wu, 2016. Assessment of the key aroma compounds in rose-based products. J. Food Drug Anal., 24: 471-476.
- Middha, D. and A. Negi, 2019. Forensic chemical profiling of flavouring additives in seized mu’assel (shisha) by gas chromatography-mass spectrometry (GC-MS). Egypt. J. Forensic Sci.
How to Cite this paper?
APA-7 Style
M.,
O.B., B.,
E.E. (2021). Analysis of Bioactive Compounds Present in the Leaf Extracts of Senna alata, Dennettia tripetalla and Delonix regia. Asian Journal of Emerging Research, 3(1), 59-64. https://doi.org/10.3923/ajerpk.2021.59.64
ACS Style
M.,
O.B.; B.,
E.E. Analysis of Bioactive Compounds Present in the Leaf Extracts of Senna alata, Dennettia tripetalla and Delonix regia. Asian J. Emerg. Res 2021, 3, 59-64. https://doi.org/10.3923/ajerpk.2021.59.64
AMA Style
M.
OB, B.
EE. Analysis of Bioactive Compounds Present in the Leaf Extracts of Senna alata, Dennettia tripetalla and Delonix regia. Asian Journal of Emerging Research. 2021; 3(1): 59-64. https://doi.org/10.3923/ajerpk.2021.59.64
Chicago/Turabian Style
M., Onyegeme-Okerenta, B., and Essien E. B..
2021. "Analysis of Bioactive Compounds Present in the Leaf Extracts of Senna alata, Dennettia tripetalla and Delonix regia" Asian Journal of Emerging Research 3, no. 1: 59-64. https://doi.org/10.3923/ajerpk.2021.59.64

This work is licensed under a Creative Commons Attribution 4.0 International License.



